Monographs: Pharmaceutical substances: Norethisterone enantate (Norethisteroni enantas)
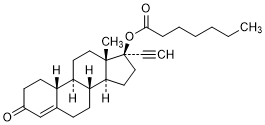
Molecular formula. C27H38O3
Relative molecular mass. 410.6
Chemical names. 3-Oxo-19-nor-17α-pregn-4-en-20-yn-17-yl heptanoate (IUPAC); 17-Hydroxy-19-nor-17α-pregn-4-en-20-yn-3-one heptanoate
CAS Reg. No. 3836-23-5.
Other name. Norethindrone enantate.
Description. A white to yellowish white, crystalline powder.
Solubility. Practically insoluble in water R; freely soluble in acetone R, methanol R or dehydrated ethanol R.
Category. Contraceptive.
Storage. Norethisterone enantate should be kept in a tightly closed container, protected from light.
Requirements
Norethisterone enantate contains not less than 97.0% and not more than 102.0% of C27H38O3, calculated with reference to the dried substance.
Identity tests
-
Either test A alone or any two of tests B, C, D or E may be applied.
-
Carry out the examination as described under 1.7 Spectrophotometry in the infrared region. The infrared absorption spectrum is concordant with the spectrum obtained from norethisterone enantate RS or with the reference spectrum of norethisterone enantate.
-
The absorption spectrum (1.6) of a 13.5 μg/mL solution of the test substances in methanol R, when observed between 210 nm and 400 nm, exhibits a maximum at about 240 nm.
Alternatively, in combination with identity test D, where a diode array detector is available, record the UV spectra of the principal peak in the chromatogram with a diode array detector in the range of 210 nm to 400 nm. The UV spectrum of the principal peak in the chromatogram obtained with solution (1) corresponds to the UV spectrum of the peak due to norethisterone enantate in the chromatogram obtained with solution (2).
-
C. Carry out the test as described under 1.14.1 Chromatography, Thin-layer chromatography using silica gel R6 or equivalent as the coating substance and a mixture of 2 volumes of cyclohexane R and 1 volume of ethyl acetate R as the mobile phase. Apply separately to the plate 5 µL of each of the following two solutions in methanol R. For solution (A), prepare a solution containing 8 mg of the test substance per mL. For solution (B), prepare a solution containing 8 mg of norethisterone enantate RS per 5.0 mL. After removing the plate from the chromatographic chamber allow it to dry in air and examine the chromatogram in ultraviolet light (254 nm). Spray the plate with 4-toluenesulfonic acid/ethanol TS, heat at 120 °C for 10 minutes and examine the chromatogram in ultraviolet light (366 nm). The principal spot obtained with solution (A) corresponds in position, appearance and intensity to that obtained with solution (B).
-
Carry out the test as described under 1.14.1 Chromatography, High-performance liquid chromatography using the conditions given under "Assay". The retention time of the principal peak in the chromatogram obtained with solution (1) corresponds to the retention time of the peak due to norethisterone enantate in the chromatogram obtained with solution (2).
-
Dissolve 1 mg of the test substance in 1 mL of dehydrated ethanol R and add 0.5 mL of sulfuric acid (~1760 g/L) TS; a violet solution is produced with a red fluorescence.
Specific optical rotation (1.4). Use a 20 mg/mL solution in dichloromethane R; 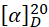 = -10.0 to -15.0.
= -10.0 to -15.0.
Sulfated ash (2.3). Not more than 1.0 mg/g.
Loss on drying. Dry over phosphorus pentoxide R at ambient temperature for 4 hours; it loses not more than 5.0 mg/g.
Related substances. Carry out the test as described under 1.14.1 Chromatography, High-performance liquid chromatography using a stainless steel column (5 cm x 4.6 mm) packed with end-capped particles of silica gel, the surface of which has been modified with chemically-bonded alkyl amide groups (1.8 μm).
Use the following conditions for gradient elution:
- Mobile phase A: mixture of water R, acetonitrile R and phosphoric acid (~1440 g/L) TS (480:520:0.01 v/v/v).
- Mobile phase B: acetonitrile R.
|
Time (min) |
Mobile phase A |
Mobile phase B |
Comments |
|
0–1.5 |
100 |
0 |
Isocratic |
|
1.5–5.5 |
100 to 67 |
0 to 33 |
Linear gradient |
|
5.5–7.5 |
67 |
33 |
Isocratic |
|
7.5–12.0 |
67 to 0 |
33 to 100 |
Linear gradient |
|
12.0–13.0 |
0 |
100 |
Isocratic |
|
13.0–13.1 |
0 to 100 |
100 to 0 |
Return to initial composition |
|
13.1–15.0 |
100 |
0 |
Re-equilibration |
Operate with a flow rate of 2.0 mL per minute. As a detector, use an ultraviolet spectrophotometer set at a wavelength of 240 nm.
Prepare the following solutions using as the diluent a mixture of 50 volumes of water R and 50 volumes of acetonitrile R. For solution (1), dissolve 65.0 mg of the test substance and dilute to 100.0 mL. For solution (2), dilute 10.0 ml of solution (1) to 100.0 mL. Dilute 1.0 ml of this solution to 100.0 mL. For solution (3), prepare a solution of norethisterone enantate for system suitability RS (containing norethisterone and impurity F) as described in the leaflet of the reference substance.
Inject 20 μL of solutions (1), (2) and (3).
The test is not valid unless the resolution between the peak due to impurity F (norethisterone caproate) and the peak due to norethisterone enantate in the chromatogram obtained with solution (3) is at least 3.0. In the chromatogram obtained with solution (2), the signal-to-noise ratio of the principal peak is at least 20.
Identify the impurities in the chromatogram obtained with solution (1) using the following relative retention with reference to norethisterone enantate (retention time about 6.4 minutes): impurity A about 0.13; impurity B about 0.54; impurity C about 0.65; impurity D about 0.75; impurity E about 0.80; impurity F about 0.86; impurity G about 1.68. Use also the chromatogram obtained with solution (3) to identify the peak impurity F (norethisterone caproate).
In the chromatogram obtained with solution (1):
-
the area of any peak corresponding to impurity E, when multiplied by a correction factor of 1.3, is not greater than 1.5 times the area of the peak due to norethisterone enantate in the chromatogram obtained with solution (2) (0.15 %);
-
the area of any impurity peak corresponding to impurities A, B, C, D, F or G is not greater than 1.5 times the area of the peak due to norethisterone enantate in the chromatogram obtained with solution (2) (0.15%);
-
the area of any other impurity peak is not greater than the area of the peak due to norethisterone enantate in the chromatogram obtained with solution (2) (0.10%).
-
The sum of the areas of all impurity peaks, including the corrected area for any peak corresponding to impurity E, is not greater than 10 times the area of the peak due to norethisterone enantate in the chromatogram obtained with solution (2) (1.0%). Disregard any peak with an area less than 0.5 times the area of the peak due to norethisterone enantate in the chromatogram obtained with solution (2) (0.05%).
Free enantic acid. Dissolve 0.3 g of the test substance in 10 mL of neutralized ethanol (~750 g/L) TS. Titrate the solution quickly with sodium hydroxide (0.01 mol/L) VS to a light blue end-point using bromothymol blue/ethanol TS as indicator; not more than 0.3 mL (corresponding to 1.3 mg/g of enantic acid).
Assay. Carry out the test under 1.14.1 Chromatography, High-performance liquid chromatography using the chromatographic conditions as described under "Related substances".
Prepare the following solutions in a mixture of acetonitrile R and water R (1:1 v/v). For solution (1), dissolve 65.0 mg of the test substance and dilute to 100.0 mL. For solution (2), dissolve 65.0 mg of norethisterone enantate RS and dilute to 100.0 mL.
Inject alternately 5 μL each of solutions (1) and (2) and record the chromatograms.
Measure the areas of the peaks corresponding to norethisterone enantate obtained in the chromatograms of solutions (1) and (2) and calculate the percentage content of norethisterone enantate (C27H38O3) using the declared content of C27H38O3 in norethisterone enantate RS.
Additional requirement for Norethisterone enantate for parenteral use
Complies with the monograph for Parenteral preparations.
Impurities
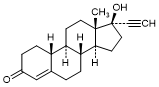
A. 17-Hydroxy-19-nor-17α-pregn-4-en-20-yn-3-one; 17α-Ethynyl-17β-hydroxy-4-estren-3-one (Norethisterone) (degradation product).
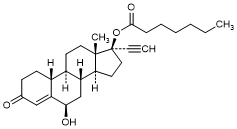
B. 6β-Hydroxy-3-oxo-19-nor-17α-pregn-4-en-20-yn-17-yl heptanoate; (6β-Hydroxy-norethisterone enantate) (degradation product).
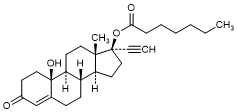
C. 10-Hydroxy-3-oxo-19-nor-17α-pregn-4-en-20-yn-17-yl heptanoate; 17α-Ethinyl-17β-heptanoyloxy-10β-hydroxy-4-estren-3-one (10β-Hydroxy-norethisterone enantate) (degradation product).
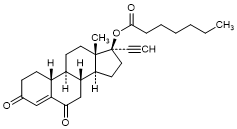
D. 3,6-Dioxo-19-nor-17α-pregn-4-en-20-yn-17-yl heptanoate (6-keto-Norethisterone enantate) (degradation product).
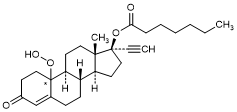
E. 10ξ-Hydroperoxy-3-oxo-19-nor-17α-pregn-4-en-20-yn-17-yl heptanoate (10-Hydroperoxy-norethisterone enantate) (degradation product).
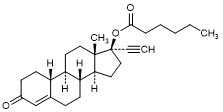
F. 3-Oxo-19-nor-17α-pregn-4-en-20-yn-17-yl hexanoate; 17α-Ethynyl-17β-hexanoyloxy-4-estren-3-one (Norethisterone caproate) (synthesis-related impurity).
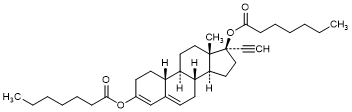
G. 19-Nor-17α-pregna-3,5-dien-20-yne-3,17-diyl diheptanoate; 17α-Ethinyl-3,17β-bis(heptanoyloxy)estra-3,5-diene (Norethisterone dienantate) (synthesis-related impurity).
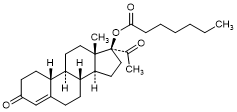
H. 3,20-Dioxo-19-nor-17α-pregn-4-en-17-yl heptanoate (17α-acetyl-norethisterone enantate) (synthesis-related impurity).
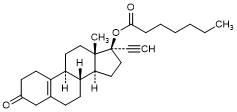
I. 3-Oxo-19-nor-17α-pregn-5(10)-en-20-yn-17-yl heptanoate (Δ5(10)-norethisterone enantate) (synthesis-related impurity).
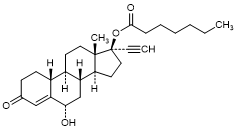
J. 6α-Hydroxy-3-oxo-19-nor-17α-pregn-4-en-20-yn-17-yl heptanoate (6α-hydroxy-norethisterone enantate) (degradation product).
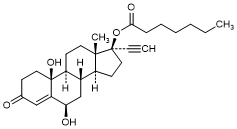
K. 6β,10β-Dihydroxy-3-oxo-19-nor-17α-pregn-4-en-20-yn-17-yl heptanoate (6β,10β-dihydroxy-norethisterone enantate) (degradation product).
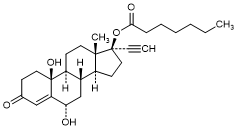
L. 6α,10β-Dihydroxy-3-oxo-19-nor-17α-pregn-4-en-20-yn-17-yl-heptanoat (6α,10β-dihydroxy-norethisterone enantate) (degradation product).