Monographs: Pharmaceutical substances: Oxamniquine (Oxamniquinum)
Molecular formula. C14H21N3O3
Relative molecular mass. 279.3
Graphic formula.
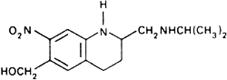
Chemical name. 1,2,3,4-Tetrahydro-2-[(isopropylamino)methyl]-7-nitro-6-quinolinemethanol; 1,2,3,4-tetrahydro-2-[[(1-methylethyl)amino]methyl]-7-nitro-6-quinolinemethanol; CAS Reg. No. 21738-42-1.
Description. A yellow-orange, crystalline powder.
Solubility. Sparingly soluble in water, soluble in methanol R and acetone R.
Category. Antischistosomal.
Storage. Oxamniquine should be kept in a well-closed container.
Requirements
Definition. Oxamniquine contains not less than 97.0% and not more than 103.0% of C14H21N3O3, calculated with reference to the anhydrous substance.
Identity tests
• Either test A or tests B and C may be applied.
A. Carry out the examination as described under 1.7 Spectrophotometry in the infrared region. The infrared absorption spectrum is concordant with the spectrum obtained from Oxamniquine RS or with the reference spectrum of oxamniquine.
B. To 10 mg add 10 mg of zinc R powder, 1.0 mL of water, and about 0.5 mL of hydrochloric acid (~250 g/l) TS. Heat in a water-bath for 5 minutes, cool in ice, add 1 mL of sodium nitrite (100 g/l) TS, and remove the excess nitrite by adding sufficient sulfamic acid (50 g/l) TS. Add 0.5 mL of the resulting solution to a mixture of 0.5 mL of 2-naphthol TS1 and 2.0 mL of sodium hydroxide (~80 g/l) TS; an orange-red colour is produced.
C. Melting temperature, about 151°C.
Heavy metals. Use 0.5 g for the preparation of the test solution as described under 2.2.3 Limit test for heavy metals, Procedure 3; determine the heavy metals content according to Method A; not more than 50 μg/g.
Iron. Ignite 1.0 g; cool and dissolve the residue in 5 mL of hydrochloric acid (~250 g/l) FeTS and 30 mL of water. Treat the solution as described under 2.2.4 Limit test for iron; the iron content is not more than 50 μg/g.
Sulfated ash. Not more than 2.0 mg/g.
Water. Determine as described under 2.8 Determination of water by the Karl Fischer method, Method A, using about 0.5 g of the substance; the water content is not more than 20 mg/g.
pH value. Shake 0.1 g with 10 mL of carbon-dioxide-free water R; pH of the suspension, 8.0-10.0.
Related substances. Carry out the test as described under 1.14.1 Chromatography, Thin-layer chromatography, using silica gel R6 as the coating substance (a precoated plate from a commercial source is suitable) and 20 volumes of chloroform R, 10 volumes of hexane R, 2 volumes of 2-propanol R, and 0.5 volume of isopropylamine R as the mobile phase. Apply separately to the plate 10 μl of each of 2 solutions in a mixture of equal volumes of chloroform R and methanol R containing (A) 25 mg of the test substance per mL and (B) 0.25 mg of the test substance per mL. After removing the plate from the chromatographic chamber, allow it to dry in air for 10 minutes until the solvents have evaporated. Examine the chromatogram in ultraviolet light first at 254 nm then at 365 nm. Any spot obtained with solution A, other than the principal spot, is not more intense than that obtained with solution B.
Assay. Dissolve about 0.3 g, accurately weighed, in 30 mL of glacial acetic acid R1, previously neutralized to oracet blue R/acetic acid TS. Titrate with perchloric acid (0.1 mol/l) VS as described under 2.6 Non-aqueous titration, Method A. Each mL of perchloric acid (0.1 mol/l) VS is equivalent to 27.93 mg of C14H21N3O3.