Monographs: Pharmaceutical substances: Oxytocin (Oxytocinum)
![]()
C43H66N12O12S2
Relative molecular mass. 1007
Chemical name. L-Cysteinyl-L-tyrosyl-L-isoleucyl-L-glutamyl-L-asparaginyl-L-cysteinyl-L-prolyl-L-leucylglycinamide cyclic (1→6)-disulfide; CAS Reg. No. 50-56-6.
Other name. Alpha-hypophamine.
Description. White or almost white powder.
Solubility. Very soluble in water. It dissolves in dilute solutions of acetic acid and of ethanol.
Category. Uterine-stimulating (Oxytocic).
Storage. Oxytocin should be kept in an airtight container, protected from light, at a temperature of 2 ºC to 8 ºC or if sterile, in a sterile, airtight, tamper-evident container.
Labelling. The label states:
- where applicable, that the substance is free from bacterial endotoxins,
- where applicable, that the substance is sterile.
Additional information. Oxytocin is hygroscopic.
Requirements
Definition. Oxytocin is a synthetic cyclic nonapeptide having the structure of the hormone produced by the posterior lobe of the pituitary gland that stimulates contraction of the uterus and milk ejection in receptive mammals. It is available in the freeze-dried form as an acetate.
Oxytocin contains not less than 93.0% and not more than 103.0% of the peptide C43H66N12O12S2, calculated with reference to the anhydrous, acetic acid-free substance.
By convention, for the purpose of labelling oxytocin preparations, 1 mg of oxytocin peptide (C43H66N12O12S2) is equivalent to 600 IU of biological activity.
Manufacture. The method of manufacture is validated to ensure that, if tested, the substance would comply with the following test:
Amino acids. Examine by means of a suitable amino-acid analyzer that has been appropriately calibrated. Standardize the apparatus with a mixture containing equimolar amounts of ammonia, glycine and the L-form of the following amino acids: lysine, threonine, alanine, leucine, histidine, serine, valine, tyrosine, arginine, glutamic acid, methionine, phenylalanine, aspartic acid, proline, isoleucine together with half the equimolar amount of L-cystine. For the validation of the method, use an appropriate internal standard, such as DL-norleucine.
Test solution Place 1.0 mg of the substance to be examined in a rigorously cleaned hard-glass tube 100 mm long and 6 mm in internal diameter. Add a suitable amount of a 50 per cent V/V solution of hydrochloric acid R. Immerse the tube in a freezing mixture at -5°C, reduce the pressure to below 133 Pa and seal. Heat at 110°C to 115 °C for 16 hours.
Cool, open the tube, transfer the contents to a 10-mL flask with the aid of five quantities, each of 0.2 mL, of water R and evaporate to dryness over potassium hydroxide R under reduced pressure. Take up the residue in water R and evaporate to dryness over potassium hydroxide R under reduced pressure; repeat these operations once. Take up the residue in a buffer solution suitable for the amino-acid analyser used and dilute to a suitable volume with the same buffer solution. Apply a suitable volume to the amino-acid analyser.
Express the content of each amino acid in moles. Calculate the relative proportions of the amino acids, taking one-sixth of the sum of the number of moles of aspartic acid, glutamic acid, proline, glycine, isoleucine and leucine as equal to one. The values fall within the following limits: aspartic acid 0.95 to 1.05; glutamic acid 0.95 to 1.05; proline 0.95 to 1.05; glycine 0.95 to 1.05; leucine 0.90 to 1.10; isoleucine 0.90 to 1.10; tyrosine 0.7 to 1.05; half-cystine 1.4 to 2.1; not more than traces of other amino acids are present.
Identity tests
• Either test A, or any two of tests B, C and D may be applied.
A. Carry out the examination as described under 1.7 Spectrophotometry in the infrared region. The infrared absorption spectrum is concordant with the spectrum obtained from oxytocin RS or with the reference spectrum of oxytocin.
B. Carry out test B.1 or, where UV detection is not available, test B.2.
B.1 Carry out the test as described under 1.14.1 Chromatography, Thin-layer chromatography, using silica gel R6 as the coating substance and a mixture of 70 volumes of dichloromethane R, 30 volumes of methanol R, 6 volumes of water R and 1 volume of glacial acetic acid R as the mobile phase. Apply separately to the plate 10 μl of each of the following two solutions in methanol R. For solution (A) use 5 mg of the test substance per mL. For solution (B) use 5 mg of oxytocin RS per mL. After removing the plate from the chromatographic chamber, allow it to dry exhaustively in a current of cool air. Examine the chromatogram in ultraviolet light (254 nm).
The principal spot obtained with solution A corresponds in position, appearance, and intensity with that obtained with solution B.
B.2 Carry out the test as described under test B.1 but using silica gel R5 as the coating substance. After removing the plate from the chromatographic chamber, allow it to dry exhaustively in a current of cool air. Spray with ninhydrin/2-propanol (5 g/l) TS. Heat the plate for a few minutes at 120°C. Examine the chromatogram in daylight.
The principal spot obtained with solution A corresponds in position, appearance, and intensity with that obtained with solution B.
C. Examine the chromatograms obtained in the Assay. The principal peak in the chromatogram obtained with the test solution is similar in retention time to the principal peak in the chromatogram obtained with the reference solution.
D. The absorption spectrum (1.6) of a 0.30 mg/mL solution, when observed between 240 nm and 330 nm, exhibits a maximum at about 275 nm; the specific absorbance (
) is 14 to 16, calculated with reference to the anhydrous, acetic acid-free substance.
Specific optical rotation (1.4). Use a 5.0 mg/mL solution and calculate with reference to the anhydrous and acetic acid-free substance; 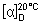 = -24.0° to -28.0°.
= -24.0° to -28.0°.
pH value (1.13). pH of a 20 mg/mL solution in carbon-dioxide-free water R, 3.0-6.0.
Water. Determine as described under 2.8 Determination of water by the Karl Fischer method, Method A, using about 0.10 g of the substance; the water content is not more than 50 mg/g.
Bacterial endotoxins. If intended for use in the manufacture of a parenteral dosage form, carry out the test as described under 3.4 Test for bacterial endotoxins; contains not more than 300 IU of endotoxin per mg.
Sterility. If intended for use in the manufacture of a parenteral dosage form without a further appropriate sterilization procedure, complies with 3.2 Test for sterility.
Acetic acid content. Carry out the test as described under 1.14.1 Chromatography, High-performance liquid chromatography, using a stainless steel column (25 cm x 4.6 mm) packed with particles of silica gel the surface of which has been modified with chemically bonded octadecylsilyl groups (5 μm).
Use the following conditions for gradient elution:
Mobile phase A: dilute 0.7 mL of phosphoric acid (~1440 g/l) TS with 900 mL of water R; adjust the pH to 3.0 with sodium hydroxide (~200 g/l) TS and dilute to 1000 mL with water R
Mobile phase B: methanol R
|
Time |
Mobile phase A |
Mobile phase B |
Comments |
|
0 – 5 |
95 |
5 |
Isocratic |
|
5 – 10 |
95 to 50 |
5 to 50 |
Linear gradient |
|
10 – 20 |
50 |
50 |
Isocratic |
|
20 – 22 |
50 to 95 |
50 to 5 |
Return to initial composition |
|
22 – 30 |
95 |
5 |
Re-equilibration |
Prepare the following solutions:
For solution (1), dissolve 15.0 mg of the substance to be examined in a mixture of 5 volumes of mobile phase B and 95 volumes of mobile phase A and dilute to 10.0 mL with the same mixture of mobile phases.
For solution (2), prepare a 0.10 g/l solution of glacial acetic acid R in a mixture of 5 volumes of mobile phase B and 95 volumes of mobile phase A.
Operate with a flow rate of 1.2 mL per minute. As a detector use an ultraviolet spectrophotometer set at a wavelength of 210 nm.
Inject alternatively 10 µl each of solutions (1) and (2). In the chromatograms obtained, the peak corresponding to acetic acid has a retention time of 3-4 minutes. The baseline presents a steep rise after the start of the linear gradient, which corresponds to the elution of oxytocin from the column. Calculate the acetic acid content; not less than 60 mg/g and not more than 100 mg/g.
Related substances. Carry out the test as described under 1.14.1 Chromatography, High-performance liquid chromatography, using the chromatographic conditions as described under Assay, with the following modifications.
Prepare the following solutions using mobile phase A as diluent. For solution (1) use 0.50 mg of the test substance per mL. For solution (2) dilute a suitable volume of solution (1) to obtain a concentration equivalent to 5.0 μg of oxytocin per mL. Prepare solution (3) using 3 mL of solution (1) and 2 mL of sulfuric acid (~10 g/l) TS, heat carefully in a boiling water-bath for 20 minutes.
Inject 50 μl of solution (3). The test is not valid unless the resolution between the peak due to oxytocin (retention time about 25 minutes) and the major peak with a relative retention of about 0.9 is at least 1.4.
Inject alternatively 50 μl each of solutions (1) and (2).
In the chromatogram obtained with solution (1), the area of any peak, other than the principal peak, is not greater than 1.5 times the area of the principal peak obtained with solution (2) (1.5%). The sum of the areas of all peaks, other than the principal peak, is not greater than 5.0 times the area of the principal peak obtained with solution (2) (5%). Disregard any peak with an area less than 0.1 times the area of the principal peak in the chromatogram obtained with solution (2) (0.1%).
Assay
Carry out the test as described under 1.14.1 Chromatography, High-performance liquid chromatography, using a stainless steel column (25 cm x 4.6 mm) packed with particles of silica gel the surface of which has been modified with chemically bonded octadecylsilyl groups (5 μm).
Use the following conditions for gradient elution:
Mobile phase A: 15 volumes of acetonitrile R, 15 volumes of phosphate buffer and 70 volumes of water R.
Mobile phase B: 70 volumes of acetonitrile R, 15 volumes of phosphate buffer and 15 volumes of water R.
Prepare the phosphate buffer by dissolving 31.2 g of sodium dihydrogen phosphate dihydrate R in 1000 mL of water R.
|
Time |
Mobile phase A |
Mobile phase B |
Comments |
|
0 – 5 |
100 |
0 |
Isocratic |
|
5 – 20 |
100 to 94 |
0 to 6 |
Linear gradient |
|
20 – 50 |
94 to 60 |
6 to 40 |
Linear gradient |
|
50 – 51 |
60 to 100 |
40 to 0 |
Return to initial composition |
|
51 – 65 |
100 |
0 |
Re-equilibration |
For solution (1) dissolve a suitable amount of the test substance, accurately weighed, in mobile phase A to obtain a solution containing 0.50 mg per mL. For solution (2) dissolve the contents of a vial of oxytocin RS in mobile phase A to obtain a concentration of 0.50 mg per mL. Prepare solution (3) using 3 mL of solution (1) and 2 mL of sulfuric acid (~10 g/l) TS, heat carefully in a boiling water-bath for 20 minutes.
Operate with a flow rate of 1.0 mL per minute. As a detector use an ultraviolet spectrophotometer set at a wavelength of 220 nm.
Maintain the column temperature at 40°C.
Inject 50 μl of solution (3). The assay is not valid unless the resolution between the peak due to oxytocin (retention time about 25 minutes) and the major peak with a relative retention of about 0.9 is at least 1.4.
Inject alternatively 50 µl each of solutions (1) and (2).
Calculate the content of oxytocin peptide (C43H66N12O12S2) from the declared content of C43H66N12O12S2 in oxytocin RS.
Impurities
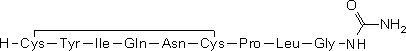
A. N-(L-cysteinyl-L-tyrosyl-L-isoleucyl-L-glutaminyl-L-asparaginyl-L-cysteinyl-L-prolyl-L-leucylglycyl)urea cyclic-(1→6)-disulfide (carbimido oxytocin),
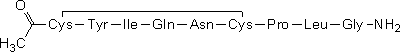
B. acetyl-L-cysteinyl-L-tyrosyl-L-isoleucyl-L-glutaminyl-L-asparaginyl-L-cysteinyl-L-prolyl-L-leucylglycinamide cyclic-(1→6)-disulfide (acetyloxytocin),
![]()
C. L-cysteinyl-L-tyrosyl-L-isoleucyl-L-glutaminyl-L-asparaginyl-L-cysteinyl-L-prolyl-L-leucylglycinamide dimer (1→1':6→6')-bisdisulfide (α-oxytocin dimer),
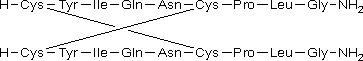
D. L-cysteinyl-L-tyrosyl-L-isoleucyl-L-glutaminyl-L-asparaginyl-L-cysteinyl-L-prolyl-L-leucylglycinamide dimer (1→6':1'→6)-bisdisulfide (β-oxytocin dimer).