Monographs: Pharmaceutical substances: Paracetamol (Paracetamolum)
Molecular formula. C8H9NO2
Relative molecular mass. 151.2
Graphic formula.
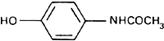
Chemical name. 4'-Hydroxyacetanilide; N-(4-hydroxyphenyl)acetamide; CAS Reg. No. 103-90-2.
Other name. Acetaminophen.
Description. A white, crystalline powder; odourless.
Solubility. Sparingly soluble in water; freely soluble in ethanol (~750 g/l) TS and acetone R; practically insoluble in ether R.
Category. Analgesic; antipyretic.
Storage. Paracetamol should be kept in a tightly closed container, protected from light.
Requirements
Definition. Paracetamol contains not less than 98.5% and not more than 101.0% of C8H9NO2, calculated with reference to the dried substance.
Identity tests
A. Dissolve 0.1 g in 100 mL of methanol R. To 1 mL of this solution add 0.5 mL of hydrochloric acid (0.1 mol/l) VS and dilute to 100 mL with methanol R. Protect the solution from light and immediately measure the absorbance of a 1-cm layer at the maximum wavelength of about 249 nm; about 0.88.
B. Dissolve 0.1 g in 10 mL of water and add 0.05 mL of ferric chloride (25 g/l) TS; a violet-blue colour is produced.
C. Boil 0.1 g with 1 mL of hydrochloric acid (~70 g/l) TS for 3 minutes, add 10 mL of water and cool; no precipitate is formed. Add 0.05 mL of potassium dichromate (0.0167 mol/l) VS; a violet colour, which does not turn to red (distinction from phenacetin) is slowly produced.
Melting range. 168-172°C.
Heavy metals. Use 1.0 g and a mixture of 85 volumes of acetone R and 15 volumes of water for the preparation of the test solution as described under 2.2.3 Limit test for heavy metals, Procedure 2; determine the heavy metals content according to Method A; not more than 10 μg/g.
Sulfated ash. Not more than 1.0 mg/g.
Loss on drying. Dry to constant weight at 105°C; it loses not more than 5.0 mg/g.
4-Aminophenol. Dissolve 0.5 g in a mixture of equal volumes of methanol R and water and dilute to 10 mL with this solvent mixture. Add 0.2 mL of alkaline sodium nitroprusside TS, mix, and allow to stand for 30 minutes. Prepare similarly a reference solution containing 0.5 g of 4-aminophenol-free paracetamol R and 0.5 mL of a solution containing 0.050 mg/mL of 4-aminophenol R in the same solvent mixture. The colour of the test solution is not more intense than that of the reference solution (0.05 mg/g).
Related substances. Carry out the test as described under 1.14.1 Chromatography, Thin-layer chromatography, using silica gel R4 as the coating substance and a mixture of 65 volumes of chloroform R, 25 volumes of acetone R, and 10 volumes of toluene R as the mobile phase. Allow the solvent front to ascend 14 cm above the line of application, using an unlined chromatographic chamber. Prepare the following 4 test solutions: For solution (A) transfer 1.0 g of finely powdered substance to be examined to a glass-stoppered tube, add 5 mL of ether R, and shake mechanically for 30 minutes. Centrifuge the tube until a clear supernatant liquid is obtained and separate this from the solid. For solution (B) dilute 1 mL of solution A to 10 mL with ethanol (~750 g/l) TS. For solution (C) dissolve 25 mg of 4-chloroacetanilide R in 50 mL of ethanol (~750 g/l) TS. For solution (D) dissolve 0.25 g of 4-chloroacetanilide R and 0.1 g of the substance to be examined in sufficient ethanol (~750 g/l) TS to produce 100 mL. Apply separately to the plate 200 μl of solution A and 40 μl of each of the remaining 3 solutions. After removing the plate from the chromatographic chamber, allow it to dry in a current of warm air and examine the chromatogram in ultraviolet light (254 nm). Any spot due to 4-chloroacetanilide obtained with solution A is not more intense than the corresponding spot obtained with solution C. Any spot obtained with solution B, other than the principal spot and the spot corresponding to 4-chloroacetanilide, is not more intense than the spot obtained with solution C. The test is valid only if the chromatogram obtained with solution D shows two distinctly separated spots corresponding to 4-chloroacetanilide and the substance being examined, the latter having a lower Rf value.
Assay. Transfer about 0.25 g, accurately weighed, to a flask, add 10 mL of hydrochloric acid (~70 g/l) TS and boil under a reflux condenser for 1 hour. Wash the condenser with 30 mL of water, add 1 g of potassium bromide R to the combined solution, and proceed as described under 2.7 Nitrite titration, titrating with sodium nitrite (0.1 mol/l) VS. Each mL of sodium nitrite (0.1 mol/l) VS is equivalent to 15.12 mg of C8H9NO2.