Monographs: Pharmaceutical substances: Penicillamine (Penicillaminum)
Molecular formula. C5H11NO2S
Relative molecular mass. 149.2
Graphic formula.
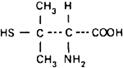
Chemical name. 3-Mercapto-D-valine; 3,3-dimethyl-D-cysteine; CAS Reg. No. 52-67-5.
Description. A white or almost white, crystalline powder; odour, characteristic.
Solubility. Soluble in 9 parts of water; slightly soluble in ethanol (~750 g/l) TS; practically insoluble in ether R.
Category. Antidote.
Storage. Penicillamine should be kept in a tightly closed container, protected from light.
Additional information. Even in the absence of light, Penicillamine is gradually degraded on exposure to a humid atmosphere, the decomposition being faster at higher temperatures.
Requirements
Definition. Penicillamine contains not less than 95.0% and not more than 100.5% of C5H11NO2S, calculated with reference to the dried substance.
Identity tests
A. Dissolve 20 mg in 4 mL of water, add 2 mL of phosphotungstic acid TS and allow to stand for a few minutes; a deep blue colour is produced.
B. Dissolve 20 mg in 5 mL of water, add 0.05 mL of sodium hydroxide (~200 g/l) TS and 20 mg of triketohydrindene hydrate R; an intense blue or violet-blue colour is produced immediately.
Specific optical rotation. Use a 50 mg/mL solution in sodium hydroxide (1 mol/l) VS; 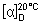 = -58° to -68°.
= -58° to -68°.
Sulfated ash. Not more than 1.0 mg/g.
Loss on drying. Dry to constant weight at 60°C under reduced pressure (not exceeding 0.6 kPa or about 5 mm of mercury); it loses not more than 5 mg/g.
pH value. pH of a 10 mg/mL solution, 4.0-6.0.
Mercury
• The operations described below must be carried out in subdued light.
Transfer about 0.5 g, accurately weighed, to a 650-mL long-necked flask containing a few glass beads, incline the flask at an angle of about 45°, and add 2.5 mL of nitric acid (~1000 g/l) TS through a small funnel placed in the mouth of the flask. Allow the mixture to stand at room temperature until nitrous oxide fumes are evolved and the vigorous reaction subsides (5-30 minutes). Add 2.5 mL of sulfuric acid (~1760 g/l) TS through the funnel and heat, gently at first and then to the production of fumes of sulfur trioxide. Cool, then add cautiously 2.5 mL of nitric acid (~1000 g/l) TS, heat again to the production of sulfur trioxide fumes, and cool. Repeat this treatment once more, then add 50 mL of water, rinsing the funnel, and collecting the rinsings in the flask. Remove the funnel, boil the solution down to approximately half its volume (about 25 mL), and cool to room temperature. Transfer to a 250-mL separating funnel with the aid of water and dilute with water to 50 mL. Add 1 mL of disodium edetate (20 g/l) TS and 1 mL of glacial acetic acid R and extract with small portions of chloroform R until the last chloroform extract remains colourless. Discard the chloroform extract and add 50 mL of sulfuric acid (0.125 mol/l) VS, 90 mL of water, and 10 mL of hydroxylamine hydrochloride (200 g/l) TS. Add dithizone standard TS, in portions of 0.3-0.5 mL, from a 10-mL burette. After each addition, shake the mixture well, allow the chloroform layer to separate, and discard it. Continue until an addition of dithizone standard TS remains green after shaking.
From the volume of the dithizone standard TS used, calculate the amount of mercury present in the test substance. It contains not more than 20 μg of Hg per g.
Assay. Dissolve about 0.1 g, accurately weighed, in 50 mL of water, add 5 mL of sodium hydroxide (1 mol/l) VS and 0.2 mL of dithizone TS and titrate with mercuric nitrate (0.02 mol/l) VS. Each mL of mercuric nitrate (0.02 mol/l) VS is equivalent to 5.968 mg of C5H11NO2S.