Monographs: Pharmaceutical substances: Pentamidine isetionate (Pentamidini isetionas)
Molecular formula. C19H24N4O2. 2 C2H6O4S
Relative molecular mass. 592.7
Graphic formula.
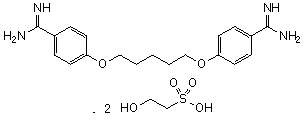
Chemical name. 4,4'-[pentane-1,5-diylbis(oxy)]dibenzenecarboximidamide bis(2-hydroxyethanesulfonate); 4,4'-(pentamethylenedioxy)dibenzamidine bis(2-hydroxyethanesulfonate); CAS Reg. No. 140-64-7.
Description. A white, or almost white, crystalline powder; odourless.
Solubility. Soluble in 10 parts of water; slightly soluble in ethanol (~750 g/L) TS; practically insoluble in ether R.
Category. Antipneumocystosis, antitoxoplasmosis and antitrypanosomal activity.
Storage. Pentamidine isetionate should be kept in a well-closed container.
Additional information. Pentamidine isetionate is hygroscopic and exhibits polymorphism.
Requirements
Definition. Pentamidine isetionate contains not less than 98.5% and not more than 102.5% of C19H24N4O2,2C2H6O4S, calculated with reference to the dried substance.
Identity tests
- Either test A alone or tests B and C may by applied.
A. Carry out the examination as described under 1.7 Spectrophotometry in the infrared region. The infrared absorption spectrum is concordant with the spectrum obtained from pentamidine isetionate RS or with the reference spectrum of pentamidine isetionate. If the spectra thus obtained are not concordant repeat the test using the residues obtained by separately dissolving the sample and pentamidine isetionate RS in a small amount of methanol R and evaporating to dryness. Dry the evaporated samples at 150–160 °C for approximately 1 hour. The infrared absorption spectrum is concordant with the spectrum obtained from pentamidine isetionate RS.
B. The absorption spectrum of a 10 μg/mL solution in hydrochloric acid (0.01 mol/L) VS, when observed between 230 nm and 350 nm, exhibits a maximum at about 262 nm; the absorbance of a 1 cm layer at this wavelength is about 470.
C. To 0.5 g add 5 mL of water and heat to 80 °C to dissolve. Add 10 mL of sodium hydroxide (~50 g/L) TS, cool in ice and filter. To 2 mL of the nitrate add 0.2 mL of nitric acid (~1000 g/L) TS followed by 0.2 mL of ceric ammonium nitrate TS; a red-orange colour is produced.
Sulfated ash. Not more than 1.0 mg/g.
Loss on drying. Dry to constant weight at 105 °C; it loses not more than 40 mg/g.
pH value (1.13). pH of a 0.05 g/mL solution, 4.5–6.5.
Related substances
Carry out the test as described under 1.14.1 Chromatography, Thin-layer chromatography using silica gel R6, activated at 105 °C for 1 hour, as the coating substance and as the mobile phase the upper layer obtained by shaking together 10 volumes of water, 8 volumes of 1-butanol R and 2 volumes of glacial acetic acid R. Apply separately to the plate 10 μL of each of 2 solutions in methanol R containing (A) 50 mg of the test substance per mL and (B) 0.25 mg of the test substance per mL. After removing the plate from the chromatographic chamber allow it to dry in air and examine the chromatogram in ultraviolet light (254 nm). Any spot obtained with solution A, other than the principal spot, is not more intense than that obtained with solution B.
Assay
Carry out Method A as described under 2.10 Determination of nitrogen using about 0.4 g of the test substance, accurately weighed, and 9 mL of nitrogen-free sulfuric acid (~1760 g/L) TS. Each mL of sulfuric acid (0.05 mol/L) VS is equivalent to 14.82 mg of C19H24N4O2,2C2H6O4S.