Monographs: Pharmaceutical substances: Pentamidine mesilate (Pentamidini mesilas)
Molecular formula. C19H24N4O2,2CH4O3S
Relative molecular mass. 532.6
Graphic formula.
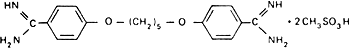
Chemical name. 4,4'-(Pentamethylenedioxy)dibenzamidine dimethanesulfonate; 4,4'-[1,5-pentanediylbis(oxy)]bis[benzenecarboximidamide] dimethanesulfonate; CAS Reg. No. 6823-79-6.
Description. A white or light pink, granular powder; almost odourless.
Solubility. Slightly soluble in water and ethanol (~750 g/l) TS; practically insoluble in ether R and acetone R.
Category. Antitrypanosomal drug; antileishmaniasis drug.
Storage. Pentamidine mesilate should be kept in a well-closed container.
Requirements
Definition. Pentamidine mesilate contains not less than 98.5% and not more than 102.5% of C19H24N4O2,2CH4O3S, calculated with reference to the dried substance.
Manufacture. The production method must be evaluated to determine the potential for formation of alkyl mesilates, which is particularly likely to occur if the reaction medium contains lower alcohols. Where necessary, the production method is validated to demonstrate that alkyl mesilates are not detectable in the final product.
Identity tests
A. To 0.5 g add 5 mL of water and heat to 80°C to dissolve. Add 10 mL of sodium hydroxide (~50 g/l) TS, cool in ice, and filter. To 2 mL of the filtrate add 0.2 mL of nitric acid (~1000 g/l) TS followed by 0.2 mL of ceric ammonium nitrate TS; a yellow colour is produced.
B. Heat 0.5 g with 1 mL of sodium hydroxide (~400 g/l) TS in a test-tube; ammonia, perceptible by its odour, is evolved.
C. Dissolve 1 g in 10 mL of water at 80°C, add 10 mL of sodium hydroxide (~50 g/l) TS, cool in ice, filter (keep the filtrate for test D), wash with 10 mL of water, and dry the precipitate at 105°C; melting temperature, about 188°C.
D. Transfer 10 mL of the filtrate obtained in test C to a platinum crucible, add 2.5 mL of hydrogen peroxide (~60 g/l) TS, mix with the help of a glass rod and evaporate to dryness on a water-bath. Dissolve the residue in 1 mL of water, add 1 mL of glacial acetic acid R and again evaporate to dryness on a water-bath, then ignite until free from carbon. Cool, mix the residue with 5 mL of water and filter, if necessary. Neutralize with hydrochloric acid (~70 g/l) TS and add an excess of 3 mL. Boil for 30 seconds, cool and proceed with reaction A described under 2.1 General identification tests as characteristic of sulfates.
Sulfated ash. Not more than 1.0 mg/g.
Loss on drying. Dry to constant weight at 105°C; it loses not more than 15 mg/g.
pH value. pH of a 0.05 g/mL solution prepared in warm water and then cooled, 4.5-6.5.
Related substances. Carry out the test as described under 1.14.1 Chromatography, Thin-layer chromatography, using silica gel R6, activated at 105°C for 1 hour, as the coating substance (a precoated plate from a commercial source is suitable), and as the mobile phase the upper layer obtained by shaking together 10 volumes of water, 8 volumes of 1-butanol R, and 2 volumes of glacial acetic acid R. Apply separately to the plate 10 μl of each of 2 solutions in methanol R containing (A) 50 mg of the test substance per mL (warm, if necessary) and (B) 0.25 mg of the test substance per mL. After removing the plate from the chromatographic chamber, allow it to dry in air and examine the chromatogram in ultraviolet light (254 nm). Any spot obtained with solution A, other than the principal spot, is not more intense than that obtained with solution B.
Assay. Carry out Method A as described under 2.10 Determination of nitrogen, using about 0.4 g, accurately weighed, and 9 mL of nitrogen-free sulfuric acid (~1760 g/l) TS. Each mL of sulfuric acid (0.05 mol/l) VS is equivalent to 13.32 mg of C19H24N4O2,2CH4O3S.