Monographs: Pharmaceutical substances: Phenobarbital (Phenobarbitalum)
Molecular formula. C12H12N2O3
Relative molecular mass. 232.2
Graphic formula.
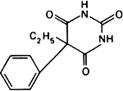
Chemical name. 5-Ethyl-5-phenylbarbituric acid; 5-ethyl-5-phenyl-2,4,6-(1H,3H,5H)-pyrimidinetrione; CAS Reg. No. 50-06-6.
Description. Colourless crystals or a white, crystalline powder; odourless.
Solubility. Soluble in about 1100 parts of water, in about 10 parts of ethanol (~750 g/l) TS and in about 15 parts of ether R.
Category. Hypnotic; sedative; anticonvulsant.
Storage. Phenobarbital should be kept in a well-closed container.
Additional information. Phenobarbital may exhibit polymorphism.
Requirements
Definition. Phenobarbital contains not less than 98.0% and not more than 101.0% of C12H12N2O3, calculated with reference to the dried substance.
Identity tests
• Either test A or tests B, C, and D may be applied.
A. Carry out the examination as described under 1.7 Spectrophotometry in the infrared region. The infrared absorption spectrum is concordant with the reference spectrum of phenobarbital.
B. Dissolve 20 mg in 5 mL of ethanol (~750 g/l) TS, add 1 drop of cobaltous chloride TS and 1 drop of ammonia (~100 g/l) TS; a violet colour is produced.
C. Shake for 3 minutes 0.1 g with 4 mL of sodium hydroxide (0.1 mol/l) VS and 1 mL of water. Filter and to 2 mL of the filtrate add 4 drops of mercuric chloride (65 g/l) TS; a white precipitate is formed, which dissolves on the addition of 5 mL of ammonia (~100 g/l) TS.
D. Dissolve 0.1 g in 2 mL of sulfuric acid (~1760 g/l) TS, add about 10 mg of sodium nitrite R, and warm on a water-bath for 10 minutes; an orange-yellow colour with a brownish sheen is produced.
Melting range. 174-178°C.
Solution in alkali. Dissolve 1.0 g in 4.0 mL of sodium hydroxide (~80 g/l) TS and add 6.0 mL of water; the solution is clear and colourless.
Sulfated ash. Not more than 1.0 mg/g.
Loss on drying. Dry to constant weight at 105°C; it loses not more than 10 mg/g.
Acidity. Boil 1.0 g with 50 mL of water for 2 minutes, adjust the volume to 50 mL, and filter. To 10 mL of the filtrate add 0.15 mL of methyl red/ethanol TS; not more than 0.1 mL of sodium hydroxide (0.1 mol/l) VS is required to obtain the midpoint of the indicator (orange).
Phenylbarbituric acid. Boil 1.0 g with 5 mL of ethanol (~750 g/l) TS for 3 minutes under a reflux condenser; a clear solution is produced.
Neutral and basic impurities. Dissolve 1.0 g in a mixture of 5 mL of sodium hydroxide (~80 g/l) TS and 10 mL of water and shake for 1 minute with 25 mL of ether R. Wash the ethereal layer 3 times, each time with 5 mL of water, evaporate the ether, and dry the residue at 105°C for 1 hour; the residue weighs not more than 3.0 mg.
Related substances. Carry out the test as described under 1.14.1 Chromatography, Thin-layer chromatography, using silica gel R2 as the coating substance and a mixture of 80 volumes of chloroform R, 15 volumes of ethanol (~750 g/l) TS, and 1 volume of ammonia (~260 g/l) TS as the mobile phase. Apply separately to the plate 10 μl of each of 2 solutions in ethanol (~750 g/l) TS containing (A) 10 mg of the test substance per mL and (B) 0.20 mg of the test substance per mL. After removing the plate from the chromatographic chamber, allow it to dry in air, and examine the chromatogram in ultraviolet light (254 nm). Any spot obtained with solution A, other than the principal spot, is not more intense than that obtained with solution B.
Assay. Dissolve about 0.20 g, accurately weighed, in 30 mL of dimethylformamide R, add 2 drops of thymolphthalein/dimethylformamide TS and titrate with sodium methoxide (0.1 mol/l) VS to a blue end-point, as described under 2.6 Non-aqueous titration. Method B. Each mL of sodium methoxide (0.1 mol/l) VS is equivalent to 23.22 mg of C12H12N2O3.