Monographs: Pharmaceutical substances: Phenytoin sodium (Phenytoinum natricum)
Molecular formula. C15H11N2NaO2
Relative molecular mass. 274.3
Graphic formula.
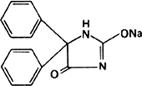
Chemical name. 5,5-Diphenylhydantoin monosodium salt; 5,5-diphenyl-2,4-imidazolidinedione monosodium salt; CAS Reg. No. 630-93-3.
Description. A white powder; odourless.
Solubility. Soluble in water, giving a slightly turbid solution owing to partial hydrolysis; soluble in ethanol (~750 g/l) TS; practically insoluble in ether R.
Category. Anticonvulsant.
Storage. Phenytoin sodium should be kept in a tightly closed container.
Additional information. Phenytoin sodium is somewhat hygroscopic and on exposure to air gradually absorbs carbon dioxide.
Requirements
Definition. Phenytoin sodium contains not less than 98.5% and not more than 101.0% of C15H11N2NaO2, calculated with reference to the dried substance.
Identity tests
• Either tests A and D or tests B, C, D, and E may be applied.
A. Shake 0.1 g with 20 mL of water, acidify with hydrochloric acid (~70 g/l) TS, and extract with chloroform R; wash the chloroform extract with water and evaporate to dryness. Carry out the examination with the residue as described under 1.7 Spectrophotometry in the infrared region. The infrared absorption spectrum is concordant with the spectrum obtained from phenytoin RS or with the reference spectrum of phenytoin.
B. Dissolve 0.1 g in a mixture of 1 mL of pyridine R and 9 mL of water, add 1 mL of copper (II) sulfate/pyridine TS, and allow to stand for 10 minutes; a blue precipitate is produced.
C. Dissolve 10 mg in 1 mL of water, add 1 drop of ammonia (~100 g/l) TS and heat until boiling begins. Add 1 drop of copper (II) sulfate/ammonia TS and shake; a pink, crystalline precipitate is formed.
D. When tested for sodium as described under 2.1 General identification tests, yields the characteristic reactions. If reaction B is to be used, prepare a 20 mg/mL solution.
E. Shake 0.1 g with 20 mL of water, acidify with hydrochloric acid (~70 g/l) TS, and extract with chloroform R; wash the chloroform extract with water and evaporate to dryness. Melting temperature of the residue, about 295°C (phenytoin).
Heavy metals. To 1.0 g add 24 mL of water and 6 mL of hydrochloric acid (~70 g/l) TS; heat the mixture until boiling begins. Filter, cool, and filter again through a suitable sintered glass filter. Dilute to 40 mL with water, mix, and determine the content of heavy metals as described under 2.2.3 Limit test for heavy metals, according to Method A; not more than 10 μg/g.
Solution in alkali. To 20 mg add 8.0 mL of carbon-dioxide-free water R and then add gradually 2.0 mL of carbonate-free sodium hydroxide (0.1 mol/L) VS; the solution is clear and not more intensely coloured than standard colour solution Yw2 when compared as described under 1.11.1 Colour of liquids.
Loss on drying. Dry to constant weight at 105°C; it loses not more than 30 mg/g.
Assay. Dissolve about 0.55 g, accurately weighed, in 30 mL of glacial acetic acid R1, add 3 drops of 1-naphtholbenzein/acetic acid TS as indicator and titrate with perchloric acid (0.1 mol/l) VS, as described under 2.6 Non-aqueous titration, Method A. Each mL of perchloric acid (0.1 mol/l) VS is equivalent to 27.43 mg of C15H11N2NaO2.
Additional requirements for Phenytoin sodium for parenteral use
Complies with the monograph for "Parenteral preparations".
Bacterial endotoxins. Carry out the test as described under 3.4 Test for bacterial endotoxins; contains not more than 0.3 IU of endotoxin RS per mg.