Monographs: Pharmaceutical substances: Phytomenadione (Phytomenadionum)
Molecular formula. C31H46O2
Relative molecular mass. 450.7
Graphic formula.
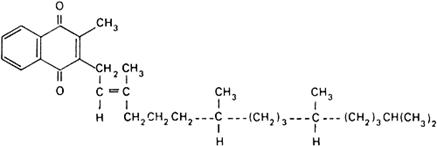
Chemical name. Phylloquinone; [R-[R*,R*-(E)]]-2-methyl-3-(3,7,11,15-tetramethyl-2-hexadecenyl)-1,4-naphthalenedione; 2-methyl-3-phytyl-1,4-naphthoquinone; CAS Reg. No. 84-80-0.
Other names. Phytonadione; Vitamin K1.
Description. A clear, yellow to amber-coloured, very viscous liquid; odourless or almost odourless.
Miscibility. Practically immiscible with water; sparingly miscible with ethanol (~750 g/l) TS; freely miscible with chloroform R and ether R.
Category. Anticoagulant.
Storage. Phytomenadione should be kept in a tightly closed container, protected from light.
Requirements
Definition. Phytomenadione contains not less than 97.0% and not more than 102.0% of C31H46O2.
Identity tests
A. The absorption spectrum of a 10 μg/mL solution in 2,2,4-trimethylpentane R, when observed between 230 nm and 350 nm, exhibits 4 maxima at about 243 nm, 249 nm, 261 nm, and 270 nm. The absorbances at those wavelengths using 1-cm cells are about 0.40, 0.42, 0.38, and 0.39, respectively. The spectrum also exhibits minima at about 246 nm, 254 nm, and 266 nm. The ratio of the absorbance at the minimum of about 254 nm to that at the maximum of about 249 nm is between 0.70 and 0.75.
B. The absorption spectrum of a 0.10 mg/mL solution in 2,2,4-trimethylpentane R, when observed between 230 nm and 350 nm, exhibits a maximum at about 327 nm and a minimum at about 285 nm. The absorbance of a 1-cm layer at the maximum is about 0.70, and at the minimum about 0.22.
C. Mix about 0.05 g of the test liquid with 5 mL of ethanol (~750 g/l) TS and add 1.0 mL of potassium hydroxide/ethanol TS1; a green colour is produced. Allow to stand for 15 minutes; the colour of the solution turns to red-brown.
Refractive index. 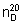 = 1.525 - 1.529.
= 1.525 - 1.529.
Acidity or alkalinity. Dissolve 1 g in 20 mL of dehydrated ethanol R; the solution is neutral to litmus paper R.
Related substances. Carry out, in subdued light, the test described under 1.14.1 Chromatography, Thin-layer chromatography, using silica gel R4 as the coating substance and a mixture of 80 volumes of cyclohexane R, 20 volumes of ether R, and 1 volume of methanol R as the mobile phase. Apply separately to the plate 10 μl of each of 2 solutions in 2,2,4-trimethylpentane R containing (A) 5 mg of the test substance per mL and (B) 0.05 mg of menadione R per mL. After removing the plate from the chromatographic chamber, allow it to dry in air and examine the chromatogram in ultraviolet light (254 nm). Any spot obtained with solution A, other than the principal spot, is not more intense than that obtained with solution B.
Assay
Carry out the following operations in subdued light.
Dissolve about 0.1 g, accurately weighed, in sufficient 2,2,4-trimethylpentane R to produce 100 mL. Dilute 10 mL to 100 mL with 2,2,4-trimethylpentane R and further dilute 10 mL of this solution to 100 mL with the same solvent. Measure the absorbance of this solution in a 1-cm layer at the maximum at about 249 nm and calculate the content of C31H46O2, using the absorptivity value of 42 (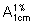 = 420).
= 420).
Additional requirements for Phytomenadione for parenteral use
Complies with the monograph for "Parenteral preparations".
Bacterial endotoxins. Carry out the test as described under 3.4 Test for bacterial endotoxins; contains not more than 14.0 IU of endotoxin RS per mg.