Monographs: Pharmaceutical substances: Pilocarpine hydrochloride (Pilocarpini hydrochloridum)
Molecular formula. C11H16N2O2,HCl
Relative molecular mass. 244.7
Graphic formula.
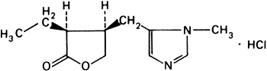
Chemical name. Pilocarpine monohydrochloride; (3S-cis)-3-ethyldihydro-4-[(1-methyl-1H-imidazol-5-yl)methyl]-2(3H)-furanone monohydrochloride; CAS Reg. No. 54-71-7.
Description. Colourless crystals or a white, crystalline powder; odourless or almost odourless.
Solubility. Very soluble in water; freely soluble in ethanol (~750 g/l) TS; insoluble in ether R.
Category. Parasympathomimetic; miotic.
Storage. Pilocarpine hydrochloride should be kept in a tightly closed container, protected from light.
Additional information. Pilocarpine hydrochloride is very poisonous; it is hygroscopic and is affected by light. Even in the absence of light, Pilocarpine hydrochloride is gradually degraded on exposure to a humid atmosphere, the decomposition being faster at higher temperatures.
Requirements
Definition. Pilocarpine hydrochloride contains not less than 98.5% and not more than 101.0% of C11H16N2O2,HCl, calculated with reference to the dried substance.
Identity tests
A. Dissolve 10 mg in 5 mL of water, add 2 drops of sulfuric acid (~100 g/l) TS, 1 mL of hydrogen peroxide (~60 g/l) TS, 1 mL of toluene R, and 1 drop of potassium dichromate (100 g/l) TS, and shake well; the toluene layer acquires a violet colour, whereas the aqueous layer remains yellow.
B. A 0.05 g/mL solution yields reaction B described under 2.1 General identification tests as characteristic of chlorides.
C. Melting temperature, about 203°C.
Specific optical rotation. Use a 50 mg/mL solution and calculate with reference to the dried substance; 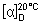 = +89° to +93°.
= +89° to +93°.
Nitrates. Dissolve 0.05 g in 5 mL of water and carefully add the solution to 5 mL of a 1 mg/mL solution of diphenylamine R in sulfuric acid (~1760 g/l) TS, ensuring that the liquids do not mix; no blue colour is produced at the interface of the two liquids.
Clarity and colour of solution. A solution of 1.0 g in 10 mL of water is clear and colourless.
Sulfated ash. Not more than 3.0 mg/g.
Loss on drying. Dry to constant weight at 105°C; it loses not more than 20 mg/g.
pH value. pH of a 5.0 mg/mL solution, 3.8-5.2.
Related alkaloids. Carry out the tests as described under 1.14.1 Chromatography, Thin-layer chromatography, using silica gel R1 as the coating substance and a mixture of 25 volumes of chloroform R, 20 volumes of acetone R, and 0.4 volumes of ammonia (~260 g/l) TS as the mobile phase. Apply separately to the plate 5 μl of each of 2 solutions containing (A) 50 mg of the test substance per mL and (B) 1.0 mg of the test substance per mL. After removing the plate from the chromatographic chamber, allow it to dry in air, spray with potassium iodobismuthate TS2, and examine the chromatogram in daylight. Any spot obtained with solution A, other than the principal spot, is not more intense than that obtained with solution B.
Assay. Dissolve 0.200 g in 50 mL of dehydrated ethanol R and add 5 mL of hydrochloric acid (0.01 mol/L) VS. Carry out a potentiometric titration using sodium hydroxide (0.1 mol/L) VS, as described under 2.6 Non-aqueous titration. Read the volume added between the two points of inflexion.
1 mL of sodium hydroxide (0.1 mol/L) VS is equivalent to 24.47 mg of C11H16N2O2,HCl.
Additional requirement for Pilocarpine hydrochloride for sterile use
Complies with 3.2 Test for sterility.