Monographs: Pharmaceutical substances: Potassium citrate (Kalii citras)
Molecular formula. C6H5K3O7,H2O
Relative molecular mass. 324.4
Graphic formula.
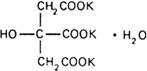
Chemical name. Tripotassium citrate monohydrate; tripotassium 2-hydroxy-1,2,3-propanetricarboxylate monohydrate; CAS Reg. No. 6100-05-6 (mono-hydrate).
Description. Transparent crystals or a white, granular powder; odourless.
Solubility. Freely soluble in water; practically insoluble in ethanol (~750 g/l) TS.
Category. Systemic alkalinizing substance; component of oral rehydration salt mixtures.
Storage. Potassium citrate should be kept in a tightly closed container.
Additional information. Potassium citrate is deliquescent when exposed to moist air.
Requirements
Definition. Potassium citrate contains not less than 99.0% and not more than 101.0% of C6H5K3O7, calculated with reference to the anhydrous substance.
Identity tests
A. To a 0.1 g/mL solution add 2 mL of sodium hydroxide (~80 g/l) TS; it yields the reaction described under 2.1 General identification tests as characteristic of potassium.
B. A 0.1 g/mL solution yields reaction A described under 2.1 General identification tests as characteristic of citrates.
Heavy metals. Use 1.0 g for the preparation of the test solution as described under 2.2.3 Limit test for heavy metals, Procedure 1; determine the heavy metals content according to Method A; not more than 10 μg/g.
Sodium. Dissolve 1 g in 10 mL of water, add 6 mL of potassium antimonate TS, and allow to stand for 15 minutes; the solution is clear or any opalescence produced is not more pronounced than that of opalescence standard TS2.
Oxalates and tartrates. Dissolve 1 g in 4 mL of hydrochloric acid (~70 g/l) TS, add 4 mL of ethanol (~750 g/l) TS and 1.0 mL of calcium chloride (55 g/l) TS; the solution remains unchanged within 1 hour.
Clarity and colour of solution. A solution of 1.0 g in 10 mL of carbon-dioxide-free water R is clear and colourless.
Readily carbonizable substances. Dissolve 0.20 g in 10 mL of sulfuric acid (~1760 g/L) TS and heat on a water-bath at 80-90°C for 1 hour; the solution is not more intensely coloured than standard colour solutions Yw5 or Gn6 when compared as described under 1.11.1 Colour of liquids.
Water. Determine as described under 2.8 Determination of water by the Karl Fischer method, Method A, using about 0.5 g of the substance; stir and allow it to remain in contact with the dehydrated methanol R for 15 minutes, stir again for 1 minute, and then titrate; the water content is not less than 40 mg/g and not more than 70 mg/g.
Acidity or alkalinity. Dissolve 1 g in 10 mL of carbon-dioxide-free water R and add 0.1 mL of phenolphthalein/ethanol TS; not more than 0.2 mL of hydrochloric acid (0.1 mol/l) VS or 0.2 mL of sodium hydroxide (0.1 mol/l) VS is required to change the colour of the solution.
Assay. Dissolve about 0.15 g, accurately weighed, in 20 mL of glacial acetic acid R1, heat to about 50°C, allow to cool to room temperature, add 0.25 mL of 1-naphtholbenzein/acetic acid TS, and titrate with perchloric acid (0.1 mol/l) VS until a green colour is obtained as described under 2.6 Non-aqueous titration, Method A. Each mL of perchloric acid (0.1 mol/l) VS is equivalent to 10.21 mg of C6H5K3O7.