Monographs: Pharmaceutical substances: Prednisolone acetate (Prednisoloni acetas)
Molecular formula. C23H30O6
Relative molecular mass. 402.5
Graphic formula.
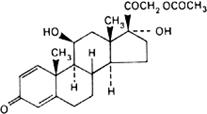
Chemical name. 11β,17,21-Trihydroxypregna-1,4-diene-3,20-dione 21-acetate; 21-(acetyloxy)-11β,17-dihydroxypregna-1,4-diene-3,20-dione; CAS Reg. No. 52-21-1.
Description. A white or almost white, crystalline powder; odourless.
Solubility. Practically insoluble in water; slightly soluble in ethanol (~750 g/l) TS and acetone R.
Category. Adrenal hormone.
Storage. Prednisolone acetate should be kept in a well-closed container, protected from light.
Additional information. Prednisolone acetate has a melting temperature of about 235°C with decomposition.
Requirements
Definition. Prednisolone acetate contains not less than 96.0% and not more than 104.0% of C23H30O6, calculated with reference to the dried substance.
Identity tests
• Either tests A and C or tests B and C may be applied.
A. Carry out the examination as described under 1.7 Spectrophotometry in the infrared region. The infrared absorption spectrum is concordant with the spectrum obtained from prednisolone acetate RS or with the reference spectrum of prednisolone acetate.
B. Carry out the test as described under 1.14.1 Chromatography, Thin-layer chromatography, using kieselguhr R1 as the coating substance and a mixture of 10 volumes of formamide R and 90 volumes of acetone R to impregnate the plate, dipping it about 5 mm into the liquid. After the solvent has reached a height of at least 16 cm, remove the plate from the chromatographic chamber and allow it to stand at room temperature until the solvents have completely evaporated. Use the impregnated plate within 2 hours, carrying out the chromatography in the same direction as the impregnation. Use 75 volumes of toluene R and 25 volumes of chloroform R as the mobile phase. Apply separately to the plate 2 μl of each of 2 solutions in a mixture of 9 volumes of chloroform R and 1 volume of methanol R containing (A) 2.5 mg of the test substance per mL and (B) 2.5 mg of prednisolone acetate RS per mL. Develop the plate for a distance of 15 cm. After removing the plate from the chromatographic chamber, allow it to dry in air until the solvents have evaporated, heat it at 120°C for 15 minutes, spray it with a mixture of 20 mL of sulfuric acid (~190 g/l) TS and 80 mL of ethanol (~750 g/l) TS, and then heat it at 120°C for 10 minutes. Allow it to cool and examine the chromatogram in daylight and in ultraviolet light (365 nm). The principal spot obtained with solution A corresponds in position, appearance, and intensity with that obtained with solution B.
C. To 0.05 g add 2 mL of ethanol (~750 g/l) TS and 2 mL of sulfuric acid (~700 g/l) TS and boil gently for 1 minute; ethyl acetate, perceptible by its odour (proceed with caution), is produced.
Specific optical rotation. Use a 10 mg/mL solution in dioxan R; 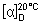 = +112° to +119°.
= +112° to +119°.
Loss on drying. Dry to constant weight at 105°C; it loses not more than 10 mg/g.
Related substances. Carry out the test as described under 1.14.1 Chromatography, Thin-layer chromatography, using silica gel R1 as the coating substance and a mixture of 95 volumes of dichloroethane R, 5 volumes of methanol R, and 0.2 volumes of water as the mobile phase. Apply separately to the plate 1 μl of each of 2 solutions in a mixture of 9 volumes of chloroform R and 1 volume of methanol R containing (A) 15 mg of the test substance per mL and (B) 0.30 mg of the test substance per mL. After removing the plate from the chromatographic chamber, allow it to dry in air until the solvents have evaporated, then heat it at 105°C for 10 minutes. Allow it to cool, spray it with blue tetrazolium/sodium hydroxide TS, and examine the chromatogram in daylight. Any spot obtained with solution A, other than the principal spot, is not more intense than that obtained with solution B.
Assay. Dissolve about 20 mg, accurately weighed, in sufficient methanol R to produce 100 mL; dilute 5.0 mL of this solution to 100 mL with the same solvent. Measure the absorbance of a 1-cm layer of the diluted solution at the maximum at about 243 nm. Calculate the amount of C23H30O6 in the substance being tested by comparison with prednisolone acetate RS, similarly and concurrently examined. In an adequately calibrated spectrophotometer the absorbance of the reference solution should be 0.37 ± 0.02.