Monographs: Pharmaceutical substances: Procarbazine hydrochloride (Procarbazini hydrochloridum)
Molecular formula. C12H19N3O,HCl
Relative molecular mass. 257.8
Graphic formula.
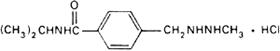
Chemical name. N-Isopropyl-α-(2-methylhydrazino)-p-toluamide monohydrochloride; N-(1-methylethyl)-4-[(2-methylhydrazino)methyl]benzamide mono-hydrochloride; CAS Reg. No. 366-70-1.
Description. A white to yellowish, crystalline powder.
Solubility. Soluble in water and methanol R; sparingly soluble in ethanol (~750 g/l) TS; practically insoluble in ether R.
Category. Cytotoxic drug.
Storage. Procarbazine hydrochloride should be kept in a tightly closed container, protected from light.
Additional information. Procarbazine hydrochloride melts at about 223°C with decomposition. Even in the absence of light, Procarbazine hydrochloride is gradually degraded on exposure to a humid atmosphere, the decomposition being faster at higher temperatures. CAUTION: Procarbazine hydrochloride must be handled with care, avoiding contact with the skin and inhalation of airborne particles. Wear rubber gloves while handling this substance.
Requirements
Definition. Procarbazine hydrochloride contains not less than 98.5% and not more than 100.5% of C12H19N3O,HCl, calculated with reference to the dried substance.
Identity tests
A. Carry out the examination as described under 1.7 Spectrophotometry in the infrared region. The infrared absorption spectrum is concordant with the spectrum obtained from procarbazine hydrochloride RS or with the reference spectrum of procarbazine hydrochloride.
B. A 0.10 g/mL solution yields reaction B described under 2.1 General identification tests as characteristic of chlorides.
Heavy metals. Use 1.0 g for the preparation of the test solution as described under 2.2.3 Limit test for heavy metals, Procedure 1; determine the heavy metals content according to Method A; not more than 20 μg/g.
Sulfated ash. Not more than 1.0 mg/g.
Loss on drying. Dry to constant weight at 105°C; it loses not more than 5.0 mg/g.
pH value. pH of a 0.05 g/mL solution, 3.0-4.5.
Assay. Dissolve 0.200 g in 100 mL of water R. Carry out a potentiometric titration using sodium hydroxide (0.1 mol/L) VS.
1 mL of sodium hydroxide (0.1 mol/L) VS is equivalent to 25.78 mg of C12H19N3O,HCl.