Monographs: Pharmaceutical substances: Proguanil hydrochloride (Proguanili hydrochloridum)
C11H16ClN5,HCl
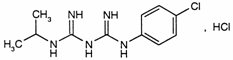
Relative molecular mass. 290.2
Chemical name. 1-(p-Chlorophenyl)-5-isopropylbiguanide hydrochloride; CAS Reg. No. 637-32-1.
Description. A white, crystalline powder.
Solubility. Slightly soluble in water, more soluble in hot water; sparingly soluble in ethanol (~750 g/l) TS.
Category. Antimalarial drug.
Storage. Proguanil hydrochloride should be kept in a well-closed container, protected from light.
Requirements
Proguanil hydrochloride contains not less than 99.0% and not more than 101.0% of C11H16ClN5,HCl, calculated with reference to the dried substance.
Identity tests
• Either tests A and D or tests B, C, and D may be applied.
A. Carry out the examination as described under 1.7 Spectrophotometry in the infrared region. The infrared absorption spectrum is concordant with the spectrum obtained from proguanil hydrochloride RS or with the reference spectrum of proguanil hydrochloride.
B. Dissolve about 0.1 g in 10 mL of water, add 5 drops of potassium ferrocyanide (45 g/l) TS; a white precipitate is produced. Add 10 - 15 drops of nitric acid (~130 g/l) TS; the precipitate dissolves.
C. Dissolve about 0.1 g in 10 mL of water, add 3 drops of copper(II) sulfate (160 g/l) TS and 1.0 mL of ammonia (~100 g/l) TS, shake, add 5 mL of toluene R, and shake again; a violet colour is produced in the toluene layer.
D. A 20mg/mL solution yields reaction A described under 2.1 General identification tests as characteristic of chlorides.
Sulfated ash. Not more than 1.0mg/g
Loss on drying. Dry to constant mass at 105 °C; it loses not more than 5.0 mg/g.
Acidity or alkalinity. To 35 mL of water maintained at a temperature of about 65°C add 0.20 mL of methyl red/methylthioninium chloride TS, neutralize with sodium hydroxide (0.01 mol/l) VS or hydrochloric acid (0.01 mol/l) VS, add 0.4 g of Proguanil hydrochloride, and stir until dissolved; the resulting solution is not acidic and requires for neutralization not more than 0.2 mL of hydrochloric acid (0.01 mol/l) VS.
Chloraniline. For solution A, dissolve 0.10 g in 1 mL of hydrochloric acid (~70 g/l) TS, and add sufficient water to produce 20 mL. Cool to 5 °C, add 1ml of sodium nitrite (35 g/l) TS, allow to stand at 5 °C for 5 minutes, add 2 mL of ammonium sulfamate (50g/l) TS, shake, and allow to stand for 10 minutes. Add 2ml of freshly prepared N-(1-naphthyl)ethylenediamine hydrochloride (1 g/l) TS, dilute to 50 mL with water, and allow to stand for 30 minutes. For solution B, treat similarly 20 mL of a solution containing 1.25μg of chloraniline R per mL. Any magenta colour produced in solution A is not more intense than that produced in solution B (250μg/g).
Related substances. Carry out the test as described under 1.14.1 Chromatography, High-performance liquid chromatography, using a stainless steel column (10cm × 5.0mm) packed with particles of silica gel, the surface of which has been modified with chemically bonded octadecylsilyl groups (5μm). As the mobile phase, use a solution prepared as follows: dissolve 1.88 g of sodium hexanesulfonate R in 1000 mL of a mixture of 12 volumes of methanol R, 8 volumes of water, and 0.1 volume of glacial acetic acid R.
Prepare the following solutions in the mobile phase: solution (A) 1.0μg of Proguanil hydrochloride per mL; and solution (B) 0.10mg of Proguanil hydrochloride per mL.
Operate with a flow rate of 1.0 mL per minute. As a detector use an ultraviolet spectrophotometer set at a wavelength of about 254nm.
Inject alternately 10μl each of solutions A and B.
Measure the areas of the peak responses obtained in the chromatograms from solutions A and B, and calculate the content of the related substances as a percentage. The sum of the areas of any peaks, other than the principal peak, in the chromatogram obtained from solution B is not greater than that of the principal peak obtained with solution A (1%).
Assay. Suspend 0.100 g in 20 mL of anhydrous acetic acid R, shake and heat at 50 °C for 5 minutes. Cool to room temperature and add 40 mL of acetic anhydride R.
In order to avoid overheating in the reaction medium, mix thoroughly throughout the titration and stop the titration immediately after the end-point has been reached.
Carry out a potentiometric titration using perchloric acid (0.1 mol/L) VS, as described under 2.6 Non-aqueous titration.
1 mL of perchloric acid (0.1 mol/L) is equivalent to 14.51 mg of C11H16ClN5,HCl.