Monographs: Pharmaceutical substances: Benzathine benzylpenicillin (Benzathini benzylpenicillinum)
Benzathine benzylpenicillin (non-injectable)
Benzathine benzylpenicillin, sterile
Molecular formula. (C16H18N2O4S)2,C16H20N2 (anhydrous)
Relative molecular mass. 909.1 (anhydrous)
Graphic formula.
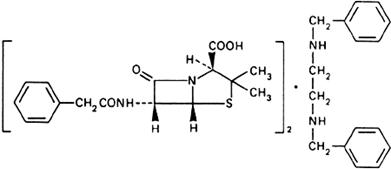
Chemical name. N,N'-Dibenzylethylenediamine compound with (2S,5R,6R)-3,3-dimethyl-7-oxo-6-(2-phenylacetamido)-4-thia-1-azabicyclo[3.2.0]heptane-2-carboxylic acid (1:2); N,N'-bis(phenylmethyl)-1,2-ethanediamine compound with [2S-(2α,5α,6β)]-3,3-dimethyl-7-oxo-6-[(phenylacetyl)amino]-4-thia-1-azabicyclo[3.2.0]heptane-2-carboxylic acid (1:2); N,N'-dibenzylethylenediamine salt of benzylpenicillin; CAS Reg. No. 1538-09-6 (anhydrous).
Other name. Penicillin G benzathine.
Description. A white powder; odourless or almost odourless.
Solubility. Very slightly soluble in water; sparingly soluble in ethanol (~750 g/l) TS; practically insoluble in ether R.
Category. Antibacterial drug.
Storage. Benzathine benzylpenicillin should be kept in a tightly closed container, protected from light, and stored at a temperature not exceeding 30 °C.
Labelling. The designation sterile Benzathine benzylpenicillin indicates that the substance complies with the additional requirements for sterile Benzathine benzylpenicillin and may be used for parenteral administration or for other sterile applications.
Additional information. Benzathine benzylpenicillin contains a variable amount of water of crystallization.
Requirements
Definition. Benzathine benzylpenicillin contains not less than 96.0% and not more than 100.5% of total penicillins calculated as (C16H18N2O4S)2,C16H20N2, and not less than 24.0% and not more than 27.0% of C16H20N2, both calculated with reference to the anhydrous substance.
Identity tests
A. To 2 mg in a test-tube add 1 drop of water followed by 2 mL of sulfuric acid (~1760 g/l) TS and mix; the solution is almost colourless. Immerse the test-tube for 1 minute in a water-bath; the solution remains almost colourless. Place 2 mg in a second test-tube, add 1 drop of water and 2 mL of formaldehyde/sulfuric acid TS and mix; the solution is almost colourless and after a few minutes the colour changes to yellow-brown. Immerse the test-tube for 1 minute in a water-bath; a reddish brown colour is produced.
B. Shake 0.1 g with 2 mL of sodium hydroxide (1 mol/l) VS for 2 minutes, extract the mixture with 2 quantities, each of 3 mL of ether R, evaporate the combined extracts, and dissolve the residue in 1 mL of ethanol (~375 g/l) TS. Add 5 mL of trinitrophenol (7 g/l) TS, heat at 90 °C for 5 minutes, and allow to cool slowly. Collect the precipitate and recrystallize it from hot ethanol (~150 g/l) TS that contains 10 mg/mL of trinitrophenol R; melting temperature, about 214 °C (picrate).
Water. Determine as described under 2.8 Determination of water by the Karl Fischer method, Method A, using about 0.5 g of the substance; the water content is not less than 50 mg/g and not more than 80 mg/g.
pH value. pH of a saturated solution containing about 0.05 g in 10 mL of carbon-dioxide-free water R, 5.0-7.5.
Assay
A. For total penicillins. Dissolve about 0.065 g, accurately weighed, in 10 mL of dimethylformamide R and dilute with sufficient water to produce 1000 mL. Transfer two 2.0-mL aliquots of this solution into separate stoppered tubes. To one tube add 10.0 mL of imidazole/mercuric chloride TS, mix, stopper the tube and place it in a water-bath at 60 °C for exactly 25 minutes. Cool the tube rapidly to 20 °C (solution A).
To the second tube add 10.0 mL of water and mix (solution B).
Without delay measure the absorbance of a 1-cm layer at the maximum at about 325 nm against a solvent cell containing a mixture of 2.0 mL of water and 10.0 mL of imidazole/mercuric chloride TS for solution A and water for solution B.
From the difference between the absorbance of solution A and that of solution B, calculate the amount of (C16H18N2O4S)2,C16H20N2 in the substance being tested by comparison with 0.050 g of benzylpenicillin sodium RS similarly and concurrently examined, taking into account that each mg of benzylpenicillin sodium RS (C16H17N2O4S) is equivalent to 1.275 mg of (C16H18N2O4S)2,C16H20N2. In an adequately calibrated spectrophotometer, the absorbance of the reference solution should be 0.62 ± 0.03.
B. For C16H20N2. To about 1 g, accurately weighed, add 30 mL of sodium chloride (400 g/l) TS and 10 mL of sodium hydroxide (~150 g/l) TS, shake well, and extract with four quantities, each of 50 mL of ether R. Wash the combined extracts with three quantities, each of 10 mL of water, extract the combined washings with 25 mL of ether R, and add the extract to the main ether solution. Evaporate the ether solution to a low bulk, add 2 mL of dehydrated ethanol R and evaporate to dryness. To the residue add 50 mL of glacial acetic acid R and titrate with perchloric acid (0.1 mol/l) VS, using 1 mL of 1-naphtholbenzein/acetic acid TS as indicator. Repeat the operation without the substance being examined; the difference between the titrations represents the amount of perchloric acid required to neutralize the liberated base. Each mL of perchloric acid (0.1 mol/l) VS is equivalent to 12.02 mg of C16H20N2.
Additional Requirements for Benzathine Benzylpenicillin for sterile use
Storage. Sterile Benzathine benzylpenicillin should be kept in a hermetically closed container, protected from light.
Bacterial endotoxins. Carry out the test as described under 3.4 Test for bacterial endotoxins; contains not more than 0.01 IU of endotoxin RS per mg of benzylpenicillin.
Sterility. Complies with 3.2 Test for sterility.
Additional requirements for Benzathine benzylpenicillin for parenteral use
Complies with the monograph for "Parenteral preparations".