Reagents, test solutions and volumetric solutions: M - Macrogol 1000 R.... Morpholine R
Macrogol 1000 R
Description. A white, waxy mass.
Viscosity. At 100 °C, about 17.3 mm2s-1.
Macrogol 200 R
Description. A clear, colourless or almost colourless viscous liquid.
Solubility. Very soluble in acetone R and in ethanol (~750 g/L) TS; practically insoluble in ether R and in fatty oils.
Macrogol 20000 2-nitroterephthalate R
Macrogol 20000 R modified by treating with 2-nitroterephthalate acid.
Description. A hard, white or almost white, waxy solid.
Solubility. Soluble in acetone
Macrogol 20000 R
Description. White or almost white solid with a waxy or paraffin-like appearance.
Solubility. Very soluble in water, soluble in methylene chloride, practically insoluble in alcohol, in fatty oils and in mineral oils.
Macrogol 200 TS
Procedure. Pour 500 mL of macrogol 200 R into a 1000 mL, round-bottom flask. Evaporate any volatile components using a rotation evaporator. Heat to 60 °C and apply a vacuum with a pressure of 1.5–2.5 kPa for 6 hours.
Macrogol 20M R
Polyethylene glycol 20 000. A suitable grade to be used in gas-liquid chromatography.
Macrogol 400 R
Polyethylene glycol 400. Macrogol 400 R is a polymer of ethylene oxide and water, represented by the formula H(OCH2CH2) nOH, in which the average value of n lies between 8.2 and 9.1.
Description. Clear colourless (or practically colourless) viscous liquid having a slight characteristic odour; slightly hygroscopic.
Average molecular weight. Transfer to a pressure flask 2.1 g of macrogol 400 R, accurately weighed, and 25.0 mL of phthalic anhydride/pyridine TS. Insert the stopper in the flask, wrap the flask securely with cloth and immerse it in a water-bath maintained at 96–100 °C to the same depth as the mixture in the flask for 1 hour. Remove the flask retaining the cloth wrapping and allow to cool in air to room temperature. To the contents of the flask add 50 mL of carbonate-free sodium hydroxide (0.5 mol/L) VS and 5 drops of phenolphthalein/pyridine TS. Titrate with carbonate-free sodium hydroxide (0.5 mol/L) VS to a pink end-point that remains for not less than 15 seconds. Perform a blank determination in a similar manner. Calculate the average molecular weight by multiplying by 4000 the weight, in g, of the test substance and dividing the result by the difference between the volume, in mL, of carbonate-free sodium hydroxide (0.5 mol/L) VS consumed for the test substance and the blank determination. The average molecular weight is between 380 and 420.
Mass density (ρ20). 1.110–1.140 kg/L.
Congealing point. Between 4 and 8 °C, the congealing point being the average of 4 consecutive temperature readings, the highest and lowest of which differ by not more than 0.4 °C.
pH value. Between 4.5 and 7.5, in a 50 g/L solution.
Acidity or alkalinity. Dissolve 5.0 g in 50 mL of water. Add a few drops of phenol red/ethanol TS. If the solution turns yellow titrate with sodium hydroxide (0.01 mol/L) VS; if the solution turns red titrate with hydrochloric acid (0.01 mol/L) VS. Not more than 2.0 mL of titrant should be required in either case.
Sulfated ash. Not more than 10 mg/g.
Heavy metals. Mix 4 g, accurately weighed, with 1 mL of hydrochloric acid (~70 g/L) TS and dilute with water to 25 mL. The limit is 50 μg/g.
Limit of monoethylene and diethylene glycols. Dissolve 50 g in 75 mL of diphenyl ether R in a 250 mL distillation flask. Slowly distil at a pressure of 100–250 Pa (1–2 mmHg) into a receiver that is graduated to 100 mL in 1 mL subdivisions, until 25 mL of distillate have been collected. Add 25.0 mL of water to the distillate, shake the receiving flask vigorously and allow the layers to separate. Cool the container in an ice-bath to solidify and facilitate the removal of the layer of diphenyl ether R. Filter the water layer through filter-paper into a glass-stoppered, 50 mL graduated cylinder. To the filtrate add an equal volume of freshly distilled acetonitrile R and shake the cylinder until solution is complete. Pipette 10 mL of the solution into 15 mL of ceric ammonium nitrate TS, mix and within 2–5 minutes determine the absorbance of the resulting solution at about 525 nm. Use a blank consisting of 15 mL of ceric ammonium nitrate TS and 10 mL of acetonitrile (400 g/L) TS. Prepare a standard solution by mixing 10 mL of acetronile (400 g/L) TS, to which 30 mg of diethylene glycol R have been added and 15 mL of ceric ammonium nitrate TS and determine the absorbance within 2–5 minutes at about 525 nm using the same blank as above. The absorbance of the test solution should not exceed that of the standard solution.
Magnesium acetate R
C4H6MgO4,4H2O.
Description. Colourless crystals. Deliquescent.
Solubility. Freely soluble in water and ethanol (~750 g/L) TS.
Magnesium chloride (0.1 mol/L) VS
Procedure. Dissolve 20.5 g of magnesium chloride R in sufficient water to produce 1000 mL.
Method of standardization. Ascertain the exact concentration of the 0.1 mol/L solution, carrying out the complexometric titration of magnesium under 2.5 Complexometric titrations using 25 mL of magnesium chloride solution. Each mL of disodium edetate (0.1 mol/L) VS is equivalent to 20.33 mg of MgCl2,6H2O.
Magnesium chloride R
MgCl2,6H2O (SRIP, 1963, p. 110).
Magnesium oxide R
MgO.
Description. A white, very fine powder.
Solubility. Very slightly soluble in water; insoluble in ethanol (~750 g/L) TS.
Magnesium oxide R1
Complies with the requirements prescribed for magnesium oxide R with the following modifications:
Arsenic: maximum 2 ppm.
Heavy metals (2.2.3): maximum 10 ppm.
Iron: maximum 50 ppm.
Magnesium standard (10 μg/mL Mg) TS
Procedure. Dilute 10 mL of magnesium (0.1 mg/mL) TS with sufficient water to produce 100 mL.
Magnesium sulfate (50 g/L) TS
A solution of magnesium sulfate R containing about 50 g of MgSO4 per litre.
Magnesium sulfate R
MgSO4,7H2O (SRIP, 1963, p. 111).
Magnesium sulfate/sulfuric acid TS
Procedure. Dissolve 25 g of magnesium sulfate R in sufficient sulfuric acid (~100 g/L) TS to produce 100 mL.
Maleic acid R
C4H4O4.
Description. Colourless crystals.
Melting temperature. About 135 °C.
Manganese dioxide R
MnO2 (SRIP, 1963, p. 112).
Manganese sulfate (15 g/L) TS
Manganese sulfate R dissolved in water to contain 15.0 g/L of MnSO4.
Manganese sulfate R
MnSO4,H2O.
Description. Pale-red, slightly efflorescent crystals.
Solubility. Soluble in about 1 part of water and 0.6 part of boiling water; practically insoluble in ethanol (~750 g/L) TS.
Manganese/silver paper R
Procedure. To a mixture of equal volumes of silver nitrate (0.1 mol/L) VS and manganese sulfate (15 g/L) TS, add drop by drop sodium hydroxide (0.1 mol/L) VS until a persistent precipitate is produced and filter. Soak strips of filter-paper (Whatman No. 1 is suitable) for 15 minutes in the solution dry them at ambient temperature, protected from light and acidic or alkaline vapours. The manganese/silver paper R should be colourless.
Test for sensitivity. Place in a cylinder of about 40 mL capacity (height about 80 mm, internal diameter about 30 mm) 1.0 mL of ammonium chloride (10 μg/mL NH4) TS. Add 9 mL of water and 1 g of magnesium oxide R. Immediately stopper the flask using a polyethylene cap below which a manganese/silver paper R is placed. Swirl the solution carefully so that magnesium particles do not come into contact with the reagent paper. Keep the cylinder at 50–60 °C for 1 hour. A true grey colour is produced on the reagent paper.
Mebendazole R
Mebendazole of a suitable quality should be used.
Meglumine (100 g/L) TS
A solution of meglumine R containing about 100 g of C7H17NO5 per litre.
Note: Meglumine (100 g/L) TS must be freshly prepared.
Meglumine R
C7H17NO5. Meglumine as described in the monograph for Meglumine.
Menadione R
2-Methyl-1,4-naphthoquinone, C11H8O2.
Description. Bright yellow crystals.
Melting temperature. About 106 °C.
Mercaptoacetic acid R
(thioglycolic acid R). C2H4O2S (SRIP, 1963, p. 206).
Mercuric acetate R
C4H6HgO4 (SRIP, 1963, p. 112).
Mercuric acetate/acetic acid TS
Procedure. Dissolve 50 g of mercuric acetate R in sufficient glacial acetic acid R1 that has been neutralized, if necessary, to crystal violet/acetic acid TS with perchloric acid (0.1 mol/L) VS to produce 1000 mL.
Mercuric bromide AsTS
Procedure. Dissolve 5 g of mercuric bromide R in sufficient ethanol (~750 g/L) TS to produce 100 mL.
Mercuric bromide paper AsR
Procedure. Use smooth, white filter-paper weighing 65–120 g/m2. The thickness of the paper in mm should be approximately equal numerically to the weight expressed as above, divided by 400. Soak pieces of filter-paper, not less than 25 mm in width, in mercuric bromide AsTS, decant the superfluous liquid, suspend the paper over a non-metallic thread and allow it to dry, protected from light.
Storage. Store the mercuric bromide paper AsR in stoppered bottles in the dark.
Note: Paper that has been exposed to sunlight or to vapours of ammonia must not be used as it produces only a pale stain or no stain at all.
Mercuric bromide R
HgBr2 (SRIP, 1963, p. 113).
Mercuric chloride (2.7 g/L) TS
A solution of mercuric chloride R containing about 2.7 g of HgCl2 per litre.
Mercuric chloride (65 g/L) TS
A solution of mercuric chloride R containing about 65 g of HgCl2 per litre (approximately 0.25 mol/L).
Mercuric chloride R
HgCl2 (SRIP, 1963, p. 113).
Mercuric chloride/ethanol TS
Procedure. Dissolve 2 g of mercuric chloride R in sufficient ethanol (~375 g/L) TS to produce 100 mL.
Mercuric iodide R
Mercury diiodide; HgI2.
Description. A heavy, crystalline, scarlet-red powder; odourless.
Solubility. Slightly soluble in water; sparingly soluble in ethanol (~750 g/L) TS, acetone R and ether R; soluble in solutions containing an excess of potassium iodide R.
Storage. Mercuric iodide R should be stored protected from light.
Mercuric nitrate (0.01 mol/L) VS
Procedure. Dissolve about 3.5 g, accurately weighed, of mercuric nitrate R in a mixture of 5 mL of nitric acid (~1000 g/L) TS and 500 mL of water and dilute with water to 1000 mL.
Method of standardization. Ascertain the exact concentration of the 0.01 mol/L solution in the following manner: transfer 20.0 mL to a conical flask, add 2 mL of nitric acid (~1000 g/L) TS and 2 mL of ferric ammonium sulfate (45 g/L) TS. Cool to below 20 °C and titrate with ammonium thiocyanate (0.01 mol/L) VS to the first appearance of a permanent brownish colour.
Mercuric nitrate (0.02 mol/L) VS
Procedure. Weigh accurately about 6.85 g of mercuric nitrate R, dissolve in a mixture of 10 mL of nitric acid (~130 g/L) TS and 500 mL of water and dilute with water to 1000 mL.
Method of standardization. Ascertain the exact concentration of the 0.02 mol/L solution following the method described under mercuric nitrate (0.01 mol/L) VS.
Mercuric nitrate R
Hg(NO3)2,H2O.
Caution. Mercuric nitrate R is poisonous.
Description. A white or slightly yellow, deliquescent, crystalline powder.
Solubility. Soluble in water in the presence of a small quantity of nitric acid (~1000 g/L) TS.
Mercuric nitrate TS
Procedure. Dissolve 40 g of yellow mercuric oxide R in a mixture of 32 mL of nitric acid (~1000 g/L) TS and 15 mL of water.
Storage. Keep in a container protected from light.
Mercuric oxide, yellow, R
HgO (SRIP, 1963, p. 114).
Mercuric sulfate TS.
Procedure. Mix 5 g of yellow mercuric oxide R with 40 mL of water and, while stirring, add 20 mL of sulfuric acid (~1760 g/L) TS, then add 40 mL of water and stir until completely dissolved.
Mercury R
Hg (SRIP, 1963, p. 115).
Mercury/nitric acid TS
Procedure. Dissolve 3 mL of mercury R in 27 mL of cold fuming nitric acid R and dilute the solution with an equal volume of water.
Storage. The solution should be stored protected from light and for not more than 2 months.
Methanesulfonic acid R
Molecular formula. CH4O3S
Description. Colourless and corrosive liquid, strong irritant.
Solubility. Miscible with water.
Density (d). ~1.48.
Melting point. About 20 °C.
Methanol for chromatography R
Methanol for chromatography contains not less than 99.8% of CH3OH and complies with the following tests:
Absorbance (1.6): not more than 0.17 at 225 nm, determined using water R as compensation liquid.
Methanol R
CH3OH (SRIP, 1963, p. 117).
Methanol, dehydrated, R
Methanol R that complies with the following requirement: water, not more than 1.0 mg/g.
Methenamine R
Hexamethylenetetramine, C6H12N4. Contains not less than 99.0% of C6H12N4.
Description. Colourless crystals or a crystalline powder; odourless.
Solubility. Soluble in water and ethanol (~750 g/L) TS.
Acidity and alkalinity. Dissolve 2.5 g in 25 mL of water. To 10 mL add 3 drops of phenolphthalein/ethanol TS; a pink colour is produced which changes to red after the addition of 1 drop of carbonate-free sodium hydroxide (0.1 mol/L) VS. To a further 10 mL aliquot add 3 drops of bromothymol blue/ethanol TS; a blue colour is produced which changes to green-blue after the addition of 3 drops of hydrochloric acid (0.1 mol/L) VS.
Sulfated ash. Not more than 0.5 mg/g.
Assay. Dissolve about 1.5 g, accurately weighed, in 10 mL of water, add 50 mL of sulfuric acid (0.5 mol/L) VS and boil until the odour of formaldehyde is no longer perceptible. Titrate the excess of acid with sodium hydroxide (1 mol/L) VS using methyl red/ethanol TS as indicator. Each mL of sulfuric acid (0.5 mol/L) VS is equivalent to 35.05 mg of C6H12N4.
Methoxyphenylacetic R
Procedure. Dissolve 2.7 g of methoxyphenylacetic acid R in 6 mL of tetramethylammonium hydroxide (~100 g/L) TS and add 20 mL of dehydrated ethanol R.
Storage. Store in a polyethylene container.
Methoxyphenylacetic acid R
C9H10O3
Description. White, crystalline powder or white or almost white crystals.
Solubility. Sparingly soluble in water, freely soluble in ethanol (~750 g/L) TS.
Melting point. About 70° C.
Methyl ethyl ketone R
C11H16N2.
Description. A clear liquid.
A commercially available reagent of suitable grade.
Refractive index. 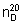 = about 1.565.
= about 1.565.
Methyl green R
[α-[p-Dimethylamino)phenyl]-α-[4-(dimethyliminio)-2,5-cyclohexadien-1-ylidene]-p-tolyl]trimethylammonium dichloride; Basic blue 20; C.I. No. 42585; C26H33Cl2N3.
Description. A green powder.
Solubility. Soluble in water; soluble in sulfuric acid (~1760 g/L) TS giving a yellow solution and turning green on dilution.
Methyl green/iodomercurate paper R
Procedure. Dip strips of suitable filter-paper in a solution of 4 g of methyl green R in 100 mL of water and allow to dry in air. Then immerse the strips for 1 hour in a mixture composed of 14 g of potassium iodide R and 20 g of mercuric iodide R in 100 mL of water. Wash the strips with water until the washings are practically colourless and allow to dry in air.
Storage. Methyl green/iodomercurate paper R should be stored protected from light.
Methyl orange ethanol TS
Procedure. Dissolve 0.04 g of methyl orange R in sufficient ethanol (~150 g/L) TS to produce 100 mL.
Methyl orange R
Sodium salt of 4'-dimethylaminoazobenzene-4-sulfonic acid, C14H14N3NaO3S (SRIP, 1963, p. 118).
Methyl orange/acetone TS
A saturated solution of methyl orange R in acetone R.
Methyl orange/ethanol TS1
Procedure . Dissolve 0.1 g of methyl orange R in 80 mL of water R and dilute to 100 mL with ethanol (~750 g/L) TS.
Test for sensitivity. A mixture of 0.1 mL of methyl orange/ethanol TS1 and 100 mL of carbon-dioxide-free water R is yellow. Not more than 0.1 mL of hydrochloric acid (1 mol/L) VS is required to change the colour to red.
Colour change. pH 3.0 (red) to pH 4.4 (yellow).
N-Methyl-N-nitrosotoluene-4-sulfonamide R
C8H10N2O3S.
Description. A yellow, crystalline powder.
Solubility. Insoluble in water; soluble in ethanol (~750 g/L) TS and ether R.
Melting temperature. About 60 °C.
3-Methylpentane-2-one R
C6H12O
Molecular weight. 100.2.
Description. Colourless, flammable liquid.
Relative density![]() . About 0.815.
. About 0.815.
Boiling point. About 118 °C.
N-Methylpiperazine R
C5H12N2.
Mass density. ρ20 = 0.902 kg/L.
Refractive index. 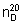 = 1.466.
= 1.466.
4(4-Methylpiperidin-1-yl)pyridine R
4(4-Methylpiperidino)pyridine ; C11H16N2.
Description. A clear liquid.
A commercially available reagent of suitable grade.
Refractive index. 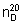 = about 1.565.
= about 1.565.
2-Methylpropanol R
C4H10O
Molecular weight. 74.1.
Other names. Isobutyl alcohol, 2-Methylpropan-1-ol.
Description. Clear colourless liquid.
Solubility. Soluble in water, miscible with ethanol (~710 g/L) TS.
Relative density![]() . About 0.80.
. About 0.80.
Boing point. About 107 °C.
Methyl red R
4'-Dimethylaminoazobenzene-2-carboxylic acid, C15H15N3O2 (SRIP, 1963, p. 118).
Methyl red/ethanol TS
Procedure. Dissolve 25 mg of methyl red R in a mixture of 0.95 mL of sodium hydroxide (0.05 mol/L) VS and 5 mL of ethanol (~750 g/L) TS, warm the solution slightly and after cooling dilute with sufficient ethanol (~375 g/L) TS to produce 250 mL.
Methyl red/methylthioninium chloride TS
Procedure. Mix 20 mL of a 0.5 mg/mL solution of methyl red R in ethanol (~150 g/L) TS with 0.4 mL of a 20 mg/mL solution of methylthioninium chloride R in water.
Methyl silicone gum R
A suitable grade to be used in gas-liquid chromatography.
2-Methylpropan-2-amine R
C4H11N
Molecular weight. 73.1.
Other names. 2-Amino-2-methylpropane, tert-Butylamine.
Description. liquid,miscible with ethanol (~710 g/L) TS.
Relative density![]() . About 0.694.
. About 0.694.
Boiling point.About 46 °C.
2-Methyl-5-nitroimidazole R
C4H5N3O2.
Melting temperature. About 253 °C.
Methyl violet 2B R
CI 42535; C.I. basic violet.
A commercially available reagent of suitable grade.
Melting point. About 137 °C, with decomposition.
Methylamine hydrochloride (20 g/L) TS
A solution of methylamine hydrochloride R containing about 20 g of CH5N,HCl per litre.
Methylamine hydrochloride R
CH5N,HCl.
Description. Deliquescent tetragonal tablets.
Solubility. Soluble in water and dehydrated ethanol R; practically insoluble in acetone R, ether R and ethyl acetate R.
Melting point. About 228 °C.
Methylisobutylketone R
Isopropylacetone, C6H12O. (SRIP, 1963, p. 119).
Methylthioninium chloride (0.2 g/L) TS
Procedure. Dissolve 23 mg of methylthioninium chloride R in sufficient water to produce 100 mL.
Methylthioninium chloride (1 g/L) TS
A solution of methylthioninium chloride R containing about 1 g of C16H18ClN3S per litre.
Methylthioninium chloride R
[methylene blue]. C16H18ClN3S,3H2O (SRIP, 1963, p. 119).
Methylthymol blue mixture R
Procedure. Mix 1 part of methylthymol blue R with 100 parts of potassium nitrate R.
Methylthymol blue R
Tetrasodium [3H-2,1-benzoxathiol-3-ylidenebis[(6-hydroxy-5-isopropyl-2-methyl-m-phenylene)methylenenitrilo]]tetraacetic acid S, S-dioxide; C37H44N2Na4O13S.
Description. A brownish-black powder.
Solubility. Freely soluble in water; very slightly soluble in ethanol (~750 g/L) TS.
Molecular sieve R
CAS Reg. No. 70955-01-0.
Molecular sieve composed of sodium aluminosilicate. It is available as beads or powder with a pore size of 0.4 nm.
When reused, it is recommended that the molecular sieve be regenerated according to the manufacturer's instructions.
Molybdenum trioxide R
MoO3 (SRIP, 1963, p. 120).
Monoethanolamine (0.1 mol/L) VS
A solution of monoethanolamine R in water to contain 6.108 g of C2H7NO in 1000 mL.
Monoethanolamine R
C2H7NO.
Description. A clear, colourless to faintly yellow, viscous liquid; odour, ammoniacal.
Miscibility. Miscible with water, methanol R and acetone R.
Boiling temperature. About 170 °C.
Mass density. ρ20= 1.01 kg/L.
Refractive index, 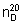 = 1.453 - 1.455.
= 1.453 - 1.455.
Mordant Black 11 indicator mixture R
Procedure. Mix 1 g of Mordant Black 11 R with 100 g of sodium chloride R.
Mordant Black 11 R
[eriochrome black R]. C.I. Mordant Black 11, C.I. No. 14645, Eriochrome Black T, Solochrome Black; sodium salt of 2-(2-hydroxy-6-nitro-4-sulfo-1-naphthylazo)-1-naphthol, C20H12N3NaO7S (SRIP, 1963, p. 84).
Morpholine R
Tetrahydro-1,4-oxazine; C4H9NO (SRIP, 1963, p. 121).