Reagents, test solutions and volumetric solutions: N - N-(1-Naphthyl)ethylenediamine hydrochloride (1 g/L) TS... 1-Nitroso-2-naphthol-3,6-disodium disulfonate R
N-(1-Naphthyl)ethylenediamine hydrochloride (1 g/L) TS
A solution of N-(1-naphthyl)ethylenediamine hydrochloride R containing about 1 g of C12H14N2,2HCl per litre.
N-(1-Naphthyl)ethylenediamine hydrochloride (5 g/L) TS
A solution of N-(1-naphthyl)ethylenediamine hydrochloride R containing about 5 g of C12H14N2,2HCl per litre.
N-(1-Naphthyl)ethylenediamine hydrochloride R
C12H14N2,2HCl (SRIP, 1963, p. 124).
N-(1-Naphthyl)ethylenediamine hydrochloride/1-propanol TS
Procedure. To 7 mL of N-(1-naphthyl)ethylenediamine hydrochloride (1 g/l) TS add 3 mL of 1-propanol R.
N-(1-Naphthyl)ethylenediamine hydrochloride/propylene glycol TS
Procedure. Dissolve 0.1 g of N-(1-naphthyl)ethylenediamine hydrochloride R in 30 mL of water and dilute to 100 mL with propylene glycol R.
Note. N -(1-Naphthyl)ethylenediamine hydrochloride/propylene glycol TS must be freshly prepared.
N,N'-bis(propan-2-yl)ethane-1,2-diamine R
C8H20N2
Molecular weight. 144.3.
Other name. N,N’-Bis(1-methylethyl)-1,2-ethanediamine; N,N’-Diisopropylethylenediamine.
Description. Colourless or yellowish, hygroscopic liquid, corrosive, flammable.
Relative density![]() . About 0.798.
. About 0.798.
Boiling point. About 170 °C.
Naphthalene-1,3-diol R
1,3-Naphthalenediol; C10H8O2.
Description. Colourless crystals.
Solubility. Freely soluble in water, ethanol (~750 g/L) TS and ether R.
Melting temperature. About 124 °C.
Naphthalene-1,3-diol/ethanol TS
Procedure. Dissolve 0.2 g of naphthalene-1,3-diol R in sufficient ethanol (~750 g/L) TS to produce 100 mL.
2-Naphthol R
[β-naphthol R] C10H8O (SRIP, 1963, p. 122).
1-Naphthol R
C10H8O.
Description. Colourless crystals or a white, crystalline powder; odour, characteristic.
Solubility. Soluble in 5 parts of ethanol (~750 g/L) TS (may form a slightly opalescent, colourless or almost colourless solution).
Melting range. 93–96 °C.
Sulfated ash. Not more than 0.5 mg/g.
1-Naphthol TS1
Procedure. Dissolve 0.10 g of 1-naphthol R in 3 mL of sodium hydroxide (~150 g/L) TS and dilute with sufficient water to produce 100 mL.
Note: 1-Naphthol TS1 must be prepared immediately before use.
2-Naphthol TS1
Procedure. Dissolve 5 g of 2-naphthol R, freshly recrystallized, in 40 mL of sodium hydroxide (~80 g/L) TS and add sufficient water to produce 100 mL.
Note: 2-Naphthol TS1 must be freshly prepared.
1-Naphtholbenzein R
C27H20O3.
Description. A reddish-brown powder.
Solubility. Practically insoluble in water; soluble in ethanol (~750 g/L) TS, benzene R, ether R and glacial acetic acid R.
1-Naphtholbenzein/acetic acid TS
Procedure. Dissolve 0.2 g of 1-naphtholbenzein R in sufficient glacial acetic acid R to produce 100 mL.
1-Naphthol/ethanol TS
Procedure. Dissolve 0.05 g of 1-naphthol R in 60 mL of ethanol (~750 g/L) TS and add sufficient water to produce 100 mL.
Neutral red R
C.I. 50040; C.I. Basic Red; C15H17ClN4 (SRIP, 1963, p. 124).
Neutral red/ethanol TS
Procedure. Dissolve 0.1 g of neutral red R in sufficient ethanol (~375 g/L) TS to produce 100 mL.
Ninhydrin/2-propanol (5g/L) TS
Procedure. Prepare a 5 g/L solution of ninhydrin R in 2-propanol R.
Ninhydrin R
See under “Triketohydrindene hydrate R”.
Nitric acid (~1000 g/L) TS
[nitric acid (70%) R] (SRIP, 1963, p. 125); d~1.41.
Nitric acid (~1000 g/L), cadmium-free and lead-free, TS
[nitric acid, cadmium-free and lead-free (70%) R].
Nitric acid (~200 g/L), cadmium-free and lead-free, TS
Procedure. Dilute 200 mL cadmium-free and lead-free nitric acid (~1000 g/L) TS with water R to produce 1000 mL.
Nitric acid (~130 g/L) TS
Procedure. Dilute 130 mL of nitric acid (~1000 g/L) TS with sufficient water to produce 1000 mL (approximately 2 mol/L); d~1.07.
Nitric acid (0.05 mol/L) VS
Nitric acid (~1000 g/L) TS diluted with water to contain 3.151 g of HNO3 in 1000 mL.
Method of standardization. Ascertain the exact concentration of the solution by following the method described under nitric acid (1 mol/L) VS.
Nitric acid (1 mol/L) VS
Nitric acid (~1000 g/L) TS diluted with water to contain 63.10 g of HNO3 in 1000 mL.
Method of standardization. Ascertain the exact concentration of the 1 mol/L solution in the following manner: Dissolve 2 g of anhydrous sodium carbonate R in 50 mL of water and titrate with the nitric acid solution using 0.1 mL of methyl orange/ethanol TS as indicator until the solution just becomes reddish yellow. Boil for 2 minutes; the solution reverts to yellow. Cool and continue the titration until the reddish yellow colour is restored. Each mL of nitric acid (1 mol/L) VS is equivalent to 0.0530 g of Na2CO3.
Nitric acid (15 g/L) TS
Nitric acid (~1000 g/L) TS diluted with water to contain 15.0 g/l of HNO3.
Nitric acid (3 g/L) TS
Nitric acid (~1000 g/L) TS diluted with water to contain 3.0 g/L of HNO3.
Nitric acid, fuming, R
HNO3 (SRIP, 1963, p. 126); d~1.5.
4-Nitroaniline R
[p-nitroaniline R] C6H6N2O2 (SRIP, 1963, p. 127).
4-Nitroaniline TS1
Procedure. Dissolve 5 g of 4-nitroaniline R in sufficient hydrochloric acid (1 mol/l) VS to produce 1000 mL.
4-Nitroaniline TS2
Procedure. Dissolve 0.4 g of 4-nitroaniline R in 60 mL of hydrochloric acid (1 mol/L) VS, cool to 15 °C and add sufficient sodium nitrite (100 g/L) TS until 1 drop of the mixture turns starch/iodine paper R blue.
Note: 4-Nitroaniline TS2 must be freshly prepared.
Nitrobenzene R
C6H5NO2 (SRIP, 1963, p. 128).
4-Nitrobenzoyl chloride R
[p-nitrobenzoyl chloride R] C7H4ClNO3 (SRIP, 1963, p. 128).
Nitrogen monoxide R
NO.
Nitric oxide, washed with water.
A commercially available gas of suitable grade.
Nitrogen monoxide and nitrogen dioxide detector tube
A cylindrical, sealed glass tube containing adsorbent filters and suitable supports for an oxidizing layer Cr(VI) salt and the diphenyl-benzidine indicator. The minimum value indicated is 5 μl/L or less, with a relative standard deviation of at most ±15%. Tubes can be verified with a calibration gas containing the appropriate impurity if a negative result is obtained.
Nitrogen R
N2 (SRIP, 1963, p. 129).
Nitrogen for chromatography R
Contains not less than 99.95% v/v of N2.
Nitrogen, oxygen-free, R
Nitrogen R which has been freed from oxygen by passing it through alkaline pyrogallol TS.
Nitromethane R
CH3NO2.
Description. A colourless, oily liquid.
Miscibility. Miscible with water, ethanol (~750 g/L) TS, ether R and di-methylformamide R.
Mass density. ρ20 = about 1.13 kg/L.
Refractive index. 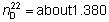 .
.
Boiling temperature. About 101 °C.
Nitrophenanthroline R
5-Nitro-1,10-phenanthroline; C12H7N3O2.
Description. A white powder; odourless.
Solubility. Soluble in water.
Melting range. 198–200 °C.
Nitrophenanthroline TS
Procedure. Dissolve 0.15 g of nitrophenanthroline R in 15 mL of freshly prepared ferrous sulfate (7 g/L) TS.
1-Nitroso-2-naphthol-3,6-disodium disulfonate (2 g/L) TS
A solution of 1-nitroso-2-naphthol-3,6-disodium disulfonate R containing about 2 g of C10H5NNa2O8S2 per litre.
1-Nitroso-2-naphthol-3,6-disodium disulfonate R
[1-nitroso-2-naphthol-3,6-disodium sulfonate R]. C10H5NNa2O8S2 (SRIP, 1963, p. 129).
1-Nitroso-4-methyl piperazine R
A commercially available substance of suitable grade.
1-Nitroso-4-methyl piperazine-d4 R
A commercially available substance of suitable grade.
N,N,N',N'-tetramethyl-2,2'-oxybis(ethaneamine) R
C8H20N2O
Molecular weight. 160.3.
Other name. Bis(2-dimethylaminoethyl) ether; 2,2’-Oxybis(N,N-dimethylethylamine).
Description. Colourless, corrosive liquid.
Relative density![]() . About 0.85.
. About 0.85.