Reagents, test solutions and volumetric solutions: P - Pancreatic digest of casein R.... Pyrogallol, alkaline, TS
Pancreatic digest of casein R
(SRIP, 1963, p. 132).
Pancreatin R
Pancreatin is a preparation of mammalian pancreas containing enzymes having protease, lipase and amylase activity. Use a commercially available reagent of suitable grade.
Storage. Pancreatin should be stored in an airtight container, protected from light at a temperature between 2° to 8 °C.
Papaic digest of soybean meal R
(SRIP, 1963, p. 134).
Paracetamol R
Paracetamol as described in the monograph for Paracetamol.
Paracetamol, 4-aminophenol-free, R
Paracetamol as described in the monograph for Paracetamol or paracetamol recrystallized from water until it complies with the following test:
Dissolve 5 g of the dried material in a mixture of equal volumes of methanol R and water and dilute to 100 mL with this solvent mixture. Add 1.0 mL of alkaline sodium nitroprusside TS, mix and allow to stand for 30 minutes; no blue or green colour is produced.
Paraffin, liquid, R
(SRIP, 1963, p. 135).
Paraformaldehyde R
(CH2O)n.
Description. A white, crystalline powder; odour, characteristic of formaldehyde.
Solubility. Slowly soluble in cold water, freely soluble in hot water, with evolution of formaldehyde; practically insoluble in ethanol (~750 g/L) TS and ether R.
Solubility in ammonia. Dissolve 1 g in 10 mL of ammonia (~100 g/L) TS; a practically clear and colourless solution is produced.
Sulfated ash. Not more than 1.0 mg/g.
Acidity or alkalinity. Shake 1 g with 20 mL of water for 1 minute and filter; the nitrate is neutral to litmus paper R.
Paraldehyde R
2,4,6-Trimethyl-s-trioxane; C6H12O3.
Description. Liquid; odour, characteristic, aromatic.
Boiling point. About 124 °C.
Mass density. ρ20 = 0.994 kg/L.
Penicillinase R
An enzyme, usually obtained from culture filtrates of a strain of Bacillus cereus, which has the specific property of inactivating penicillin by splitting the bond linking the nitrogen of the thiazolidine to the adjacent carbonyl carbon and thus releasing a carboxyl group. It is precipitated from solutions in water by acetone R, ethanol (~750 g/L) TS and dioxan R but inactivated by several hours' contact with these solvents; it is rapidly inactivated by ethyl acetate R. In place of penicillinase R a sterile filtrate obtained by fermentation of a penicillinase-producing organism in a suitable medium, described below under "Preparation of penicillinase", may also be used directly.
Description. Small, brown, easily pulverizable pieces or granules.
Solubility. Freely soluble in water, forming a slightly opalescent solution.
Preparation of penicillinase. Dissolve 10 g of pancreatic digest of casein R, 2.7 g of potassium dihydrogen phosphate R and 5.9 g of sodium citrate R in 200 mL of water, adjust the alkalinity to pH 7.2 with sodium hydroxide (~200 g/L) TS and dilute to 1000 mL with water. Dissolve 0.4 g of magnesium sulfate R in 5 mL of water and add 1 mL of ferrous ammonium sulfate (1 g/L) TS and sufficient water to produce 10 mL. Sterilize both solutions by heating in an autoclave, cool, mix, distribute in shallow layers in conical flasks and inoculate with a suitable strain (Bacillus cereus NCTC 9946 is suitable). Allow the flasks to stand at 18–37 °C until growth is apparent and then maintain at 35–37 °C for 16 hours, shaking constantly to ensure maximum aeration. Centrifuge and sterilize the supernatant liquid by filtration through a suitable membrane filter.
Penicillinase TS
A sterile aqueous solution of penicillinase R. To test the activity of penicillinase TS carry out the "Penicillinase assay". The time required for iodine decolorization is not more than 36 seconds.
Penicillinase assay. Carry out the assay in test-tubes of borosilicate glass, 15 cm long and about 20 mm in internal diameter, immersed in a water-bath of 30 ± 1 °C. All reagents should have a temperature of 30 °C before use.
To the test-tube add the reagents in the following order: 1.6 mL of gelatin TS, 0.4 mL of penicillinase TS to be tested, 1 drop of starch TS and 1 mL of benzylpenicillin sodium TS, blowing out the last reagent from a 1 mL pipette. Start the stop-watch and after 15 seconds add 2.0 mL of iodine (0.01 mol/L) VS, recording the time of decolorization of iodine from the time of the addition of benzylpenicillin sodium TS. The activity of penicillinase TS is calculated from the results of the assay. The time of decolorization of strictly 36 seconds corresponds to a penicillinase activity equivalent to a rate of decomposition (at 30 °C at pH 7.0) of 220 mg of benzylpenicillin sodium R per hour per mL of penicillinase TS.
Storage. Store between 0 and 2 °C and use within 2–3 days. When dried from the frozen state and kept in sealed ampoules, penicillinase TS may be stored for several months.
n-Pentane R
C5H12.
A commercially available reagent of suitable grade.
Description. A colourless, volatile liquid.
Boiling point. About 36 °C.
Relative density. 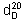 = about 1.359.
= about 1.359.
Transmittance. Not less than 20% at 200nm, 50% at 210 nm, 85% at 220 nm, 93% at 230 nm and 98% at 240 nm, determined using water in the reference cell.
1-Pentanesulfonic acid sodium salt R
C5H11NaO3S,H2O.
Description. A white, crystalline powder.
Solubility. Soluble in water.
Clarity and colour of solution. A solution of 1 g in 25 mL of water is clear and colourless.
Water. Determined by the Karl Fischer method; not more than 20 mg/g.
1-Pentanesulfonic acid TS
Procedure. Dissolve 0.960 g of 1-pentanesulfonic acid sodium salt R in 1000 mL of deaerated acetic acid (5.0 g/L) TS and adjust the pH to 4.3 with ammonia (~260 g/L) TS. Filter and deaerate before use.
Pentoxyverine citrate R
A commercially available reagent of suitable grade.
Pepsin R
Pepsin is prepared from the gastric mucosa of pigs, cattle or sheep. It contains gastric proteinases, active in acid medium (pH of 1 to 5). Use a commercially available reagent of suitable grade.
Storage. Pepsin should be stored in an airtight container protected from light at a temperature between 2° to 8 °C.]
Peptone (1 g/L) TS1
Procedure. Dissolve 1.0 g of peptone R1 (or similar peptic digest of animal tissue) in sufficient water to produce 1000 mL, filter or centrifuge to clarify, adjust the pH to 7.1 ± 0.2, place 100 mL portions into individual vessels and sterilize by maintaining at 121 °C for 18–20 minutes.
Peptone (1 g/L) TS2
Procedure. Dissolve in water, while heating, 1.0 g of dried peptone R and 9 g of sodium chloride R and dilute with sufficient water to produce 1000 mL. Adjust to pH 8.0–8.4 and boil for 20 minutes. Filter, adjust to pH 7.2–7.4 and sterilize by maintaining at 115 °C for 30 minutes.
Peptone (5 g/L) TS
Procedure. Dissolve in water, while heating, 5.0 g of dried peptone R and 7 g of sodium chloride R and dilute with sufficient water to produce 1000 mL. Adjust to pH 8.0–8.4 and boil for 20 minutes. Filter, adjust to pH 7.2–7.4 and sterilize by maintaining at 115 °C for 30 minutes.
Peptone R1
Dried peptone R that conforms to the following requirement: an autoclaved solution containing 0.02 g/mL is clear and neutral or almost neutral.
Peptone, dried, R
(SRIP, 1963, p. 137.)
Perchloric acid (~1170 g/L) TS
[perchloric acid (70%, w/w) R] (SRIP, 1963, p. 137); d~1.67.
Perchloric acid (~140 g/L) TS
Perchloric acid (~1170 g/L) TS, diluted with water to contain 141 g/L of HClO4; d~1.09.
Perchloric acid (0.02 mol/L) VS
Procedure. Dilute 20 mL of perchloric acid (0.1 mol/L) VS with sufficient glacial acetic acid R1 to produce 100 mL.
Water and method of standardization. Immediately before use determine the content of water and ascertain the exact concentration of the solution following the methods described under perchloric acid (0.1 mol/L) VS.
Perchloric acid (0.05 mol/L) VS
Procedure. To 900 mL of glacial acetic acid R1 at about 25 °C, add 4.2 mL of perchloric acid (~1170 g/L) TS, mix, add 15 mL of acetic anhydride R and mix again. Cool to room temperature, add sufficient glacial acetic acid R1 to produce 1000 mL and allow to stand for 24 hours.
Water and method of standardization. Determine the content of water and ascertain the exact concentration of the solution following the method described under perchloric acid (0.1 mol/L) VS.
Perchloric acid (0.1 mol/L) VS
Procedure. To 900 mL of glacial acetic acid R1, at about 25 °C, add 8.2 mL of perchloric acid (~1170 g/L) TS, mix, add 32 mL of acetic anhydride R and mix again. Cool to room temperature, add sufficient glacial acetic acid R1 to produce 1000 mL and allow to stand for 24 hours.
Water. Determine the content by the Karl Fischer method. If necessary add sufficient water or acetic anhydride R to adjust the content of water to between 0.1 and 2.0 mg/mL and allow to stand for a further 24 hours.
Method of standardization. Ascertain the exact concentration by titrating 0.5 g, accurately weighed, of potassium hydrogen phthalate R, previously dried at 120 °C for 2 hours, using method A, as described under 2.6 Non-aqueous titration. Each mL of perchloric acid (0.1 mol/L) VS is equivalent to 20.42 mg of C8H5KO4. Record the temperature at which the standardization is carried out.
Perchloric acid TS
Procedure. Dilute 82 mL of perchloric acid (~1170 g/L) TS with sufficient water to produce 1000 mL (approximately 1 mol/L).
Perchloric acid/dioxan (0.1 mol/L) VS
Procedure. Mix 8.5 mL of perchloric acid (~1170 g/L) TS with sufficient dioxan R, which has been especially purified by adsorption, to produce 1000 mL.
Method of standardization. Ascertain the exact concentration of the solution by titrating 0.7 g of potassium hydrogen phthalate R, accurately weighed and previously dried at 120 °C for 2 hours, using method A, as described under 2.6 Non-aqueous titration. Each mL of perchloric acid/dioxan (0.1 mol/L) VS is equivalent to 20.42 mg of C8H5KO4.
Periodic-acetic acid TS
Procedure. Dissolve 0.446 g of sodium metaperiodate R in 2.5 mL of sulfuric acid (~570 g/L) TS and dilute to 100 mL with glacial acetic acid R.
Petroleum, light, R
[light petroleum R] (SRIP, 1963, p. 108).
Petroleum, light, R1
Description. A colourless, very volatile, highly inflammable liquid.
Boiling range. 40–60 °C.
Mass density. ρ20 = 0.630–0.650 kg/L.
Petroleum, light R2
Description. Clear, colourless, flammable liquid without fluorescence.
Solubility. Practically insoluble in water, miscible with ethanol (~750 g/L) TS.
Boiling range. (1.2.4). 50–70 °C.
Mass density. ρ20 = 0.661 to 0.664 kg/L.
o-Phenanthroline (1 g/L) TS
Procedure. Dissolve 0.11 g of o-phenanthroline R in sufficient water to produce 100 mL.
o-Phenanthroline R
1,10-Phenanthroline, C12H8N2,H2O (SRIP, 1963, p. 138).
o-Phenanthroline TS
Procedure. Dissolve 0.7 g of ferrous sulfate R in about 70 mL of water, add about 1.5 g of o-phenanthroline R and sufficient water to produce 100 mL.
Phenol (50 g/L) TS
A solution of phenol R containing about 50 g of C6H6O per litre.
Phenol R
C6H6O.
Description. Colourless, or at most faintly pink, cohering or separate acicular crystals or crystalline masses; odour, characteristic. Corrosive and blanches the skin and mucous membranes.
Solubility. Soluble in about 15 parts of water and in about 100 parts of liquid paraffin R; freely soluble in ethanol (~750 g/L) TS and ether R.
Completeness of solution. 1.0 g dissolves completely in 15 mL of water at 15 °C.
Congealing temperature. Not below 40.5 °C.
Residue on evaporation. Evaporate on a water-bath and dry to constant weight at 105 °C; leaves not more than 0.5 mg/g of residue.
Phenol red R
Phenolsulfonphthalein, C19H14O5S (SRIP, 1963, p. 139).
Phenol red/ethanol TS
Procedure. Dissolve 0.05 g of phenol red R in a mixture of 2.85 mL of sodium hydroxide (0.05 mol/L) VS and 5 mL of ethanol (~710 g/L) TS. Warm the solution slightly and after cooling dilute with sufficient ethanol (~150 g/L) TS to produce 250 mL.
Phenol red/ethanol TS1
Procedure. Dissolve 0.1 g of phenol red R in a mixture of 2.82 mL of sodium hydroxide (1 mol/L) VS and 20 mL of ethanol (~750 g/L) TS and dilute to 100 mL with water R.
Test for sensitivity. Add 0.1 mL of the phenol red solution to 100 mL of carbon dioxide-free water R; the solution is yellow. Not more than 0.1 mL of sodium hydroxide (0.02 mol/L) VS is required to change the colour to reddish-violet.
Colour change. pH 6.8 (yellow) to pH 8.4 (reddish-violet).
Phenoldisulfonic acid (250 g/L) TS
A commercially available reagent of suitable grade.
Phenoldisulfonic acid TS
Description. A clear liquid which may develop a pale brown colour on storage.
Procedure. Either of the following methods of preparation can be used: (1) to 3 g of phenol R add 20 mL of sulfuric acid (~1760 g/L) TS and heat on a water-bath for 6 hours; store in a stoppered vessel; (2) dilute phenoldisulfonic acid (250 g/L) TS with sulfuric acid (~1760 g/L) TS to contain 150 g of phenol per litre.
Sensitivity to nitrate. Evaporate a solution containing 0.1 mg of potassium nitrate R to dryness in a porcelain dish on a water-bath. Cool, add 1.0 mL of the solution to be examined and allow to stand for 10 minutes. Add 10 mL of water, cool, add 10 mL of ammonia (~100 g/L) TS and dilute to 25 mL with water; a distinct yellow colour is produced when compared with a solution prepared similarly but omitting the potassium nitrate R.
Phenolphthalein R
C20H14O4 (SRIP, 1963, p. 139).
Phenolphthalein/ethanol TS
Procedure. Dissolve 1.0 g of phenolphthalein R in sufficient ethanol (~750 g/L) TS to produce 100 mL.
Phenolphthalein/ethanol TS, diluted
Procedure. Dissolve 0.1 g of phenolphthalein R in sufficient ethanol (~750 g/L) TS to produce 100 mL.
Sensitivity test. To 0.1 mL of the phenolphthalein solution add 100 mL of carbon dioxide-free water R. The solution is colourless. Not more than 0.2 mL of 0.02 M sodium hydroxide is required to change the colour to pink.
Colour change: pH 8.2 (colourless) to pH 10.0 (red).
Phenolphthalein/pyridine TS
Procedure. Dissolve 1.0 g of phenolphthalein R in sufficient pyridine R to produce 100 mL.
Phenoxyacetic acid R
Phenoxyethanoic acid; C8H8O3.
Description. White or almost white crystals.
Melting temperature. About 98 °C.
2-Phenoxyethanol R
C8H10O2.
Description. A clear, colourless, oily liquid; odour, faintly aromatic.
Miscibility. Slightly miscible with water; freely miscible with ethanol (~750 g/L) TS and ether R.
Mass density. ρ20 = about 1.1 kg/L.
Refractive index. 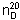 = about 1.537.
= about 1.537.
Freezing point. Not below 12.0 °C.
1,4-Phenylenediamine dihydrochloride R
C6H8N2,2HCl.
Description. A white to pale tan, crystalline powder, turning pink on exposure to air.
Solubility. Freely soluble in water; slightly soluble in ethanol (~750 g/L) TS and ether R.
Storage. Keep in a well-closed container, protected from light.
2-Phenylethanol R
C8H10O. A suitable grade to be used in gas-liquid chromatography.
2-Phenylethanol TS
Procedure. Dissolve 1 g of 2-phenylethanol R in sufficient methanol R to produce 50 mL.
Phenylhydrazine hydrochloride (10 g/L) TS
A solution of phenylhydrazine hydrochloride R containing 10 g of C6H8N2,HCl in 1000 mL.
Phenylhydrazine hydrochloride R
C6H8N2,HCl (SRIP, 1963, p. 140).
Phenylhydrazine R
C6H8N2 (SRIP, 1963, p. 140).
Phenylhydrazine/hydrochloric acid TS
Procedure. Dissolve 0.75 g of phenylhydrazine hydrochloride R in 50 mL of water and shake with 2 g of charcoal R. Filter, add 25 mL of hydrochloric acid (~420 g/L) TS and sufficient water to produce 200 mL.
Phenylhydrazine/sulfuric acid TS
Procedure. Dissolve 65 mg of phenylhydrazine hydrochloride R, previously recrystallized from ethanol (~710 g/L) TS, in a sufficient quantity of a mixture of 170 mL of sulfuric acid (~1760 g/L) TS and 80 mL of water to produce 100 mL.
Note: Phenylhydrazine/sulfuric acid TS must be freshly prepared.
Phenyl/methylpolysiloxane R
A suitable grade of a mixture to be used in gas chromatography composed of 5 g of phenylpolysiloxane and 95 g of methylpolysiloxane per 100 g.
pH-Indicator paper R
A paper impregnated with a suitable mixture of colour indicators such that the changes in colour permit estimation of the pH of a solution with an adequate sensitivity (usually 1 pH unit), at least in the pH range 1–10.
Phloroglucinol R
Benzene-1,3,5-triol dihydrate; C6H6O3,2H2O.
Description. White or pale cream coloured crystals.
Melting point. About 220 °C.
Phosphate buffer, pH 2.5, TS
Dissolve 3.48 g of dipotassium hydrogen phosphate R in 1000 mL of water R and adjust to pH 2.5 with phosphoric acid R.
Phosphate buffer, pH 3.1, TS
Dissolve 3.48 g of dipotassium hydrogen phosphate R in 1000 mL of water R and adjust to pH 3.1 with phosphoric acid R.
Phosphate buffer, pH 4.0, TS
Procedure. Dissolve 5.04 g of disodium hydrogen phosphate R and 3.01 g of potassium dihydrogen phosphate R in sufficient water to produce 1000 mL and adjust the pH to 4.0 with glacial acetic acid R.
Phosphate buffer, pH 6.4, TS
Procedure. Dissolve 1.36 g of potassium dihydrogen phosphate R in 50 mL of carbon-dioxide-free water R, add 12.60 mL of carbonate-free sodium hydroxide (0.2 mol/L) VS and dilute with sufficient carbon-dioxide-free water R to produce 200 mL.
Phosphate buffer, pH 6.9, TS
Procedure. Dissolve 3.40 g of potassium dihydrogen phosphate R and 3.55 g of disodium hydrogen phosphate R in sufficient carbon-dioxide-free water R to produce 1000 mL.
Phosphate buffer, pH 7.0 (0.01 mol/L) TS
Procedure. Dissolve 0.136 g of potassium dihydrogen phosphate R in sufficient water to produce 100 mL. Separately dissolve 2.681 g of disodium hydrogen phosphate R in sufficient water to produce 100 mL. Mix 38.9 mL of the potassium phosphate solution with 61.1 mL of the sodium phosphate solution.
Phosphate buffer, pH 7.0 (0.067 mol/L), TS
Procedure. Dissolve 0.908 g of potassium dihydrogen phosphate R in sufficient water to produce 100 mL. Separately dissolve 2.38 g of disodium hydrogen phosphate R in sufficient water to produce 100 mL. Mix 38.9 mL of the potassium phosphate solution with 61.1 mL of the sodium phosphate solution.
Phosphate buffer, pH 7.0 (0.1 mol/L) TS
Procedure. Dissolve 1.361 g of potassium dihydrogen phosphate R in 100.0 mL of water. Adjust the pH using a 14,20 g/L solution of anhydrous disodium hydrogen phosphate R.
Phosphate buffer, pH 7.0, TS
Procedure. Dissolve 5.76 g of anhydrous disodium hydrogen phosphate R and 3.55 g of potassium dihydrogen phosphate R in sufficient water to produce 1000 mL.
Phosphate buffer, pH 7.2, TS
Procedure. Dissolve 6.80 g of potassium dihydrogen phosphate R and 1.40 g of sodium hydroxide R in sufficient water to produce 1000 mL.
Phosphate buffer, pH 7.4, TS
Procedure. Dissolve 6.8 g of potassium dihydrogen phosphate R in 250 mL of water and add 393.4 mL of sodium hydroxide (0.1 mol/L) VS.
Phosphate buffer, pH 7.6, TS
Procedure. Place 1.36 g of potassium dihydrogen phosphate R in a 200 mL volumetric flask, dissolve it in water, add 42.4 mL of sodium hydroxide (0.2 mol/L) VS and dilute to volume with water.
Phosphate buffer, pH 8.0 (0.05 mol/L), TS
Procedure. Dissolve 1.361 g of potassium dihydrogen phosphate R in 50 mL of water R and add 46.8 mL of sodium hydroxide (0.2 mol/L) VS. Dilute to 200 mL with water R.
Phosphate buffer, pH 8.0, TS
Procedure. Dissolve 8.95 g of anhydrous disodium hydrogen phosphate R and 0.50 g of potassium dihydrogen phosphate R in sufficient water to produce 1000 mL.
Phosphate buffer, sterile,1 pH 10.5, TS1
Procedure. Dissolve 35.0 g of dipotassium hydrogen phosphate R in water, add 20 mL of sodium hydroxide (1 mol/L) VS and sufficient water to produce 1000 mL. If necessary adjust the pH to 10.5 with phosphoric acid (~1440 g/L) TS or potassium hydroxide (~110 g/L) TS and then sterilize the solution for 20 minutes in an autoclave at 120 °C.
Phosphate buffer, sterile,1 pH 4.5, TS
Procedure. Dissolve 13.6 g of potassium dihydrogen phosphate R in sufficient water to produce 1000 mL. If necessary adjust the pH to 4.45–4.55 with phosphoric acid (~1440 g/L) TS or potassium hydroxide (~110 g/L) TS and then sterilize the solution for 20 minutes in an autoclave at 120 °C.
Phosphate buffer, sterile,1 pH 6.0, TS1
Procedure. Dissolve 2.0 g of dipotassium hydrogen phosphate R and 8.0 g of potassium dihydrogen phosphate R in sufficient water to produce 1000 mL. If necessary adjust the pH to 5.95–6.05 with phosphoric acid (~1440 g/L) TS or potassium hydroxide (~110 g/L) TS and then sterilize the solution for 20 minutes in an autoclave at 120 °C.
Phosphate buffer, sterile,1 pH 6.0, TS2
Procedure. Dissolve 1.16 g of anhydrous disodium hydrogen phosphate R and 7.96 g of potassium dihydrogen phosphate R in sufficient water to produce 1000 mL. If necessary adjust the pH to 5.95–6.05 with phosphoric acid (~1440 g/L) TS or potassium hydroxide (~110 g/L) TS and then sterilize the solution for 20 minutes in an autoclave at 120 °C.
Phosphate buffer, sterile,1 pH 6.0, TS3
Procedure. Dissolve 20.0 g of dipotassium hydrogen phosphate R and 80.0 g of potassium dihydrogen phosphate R in sufficient water to produce 1000 mL. If necessary adjust the pH to 5.95–6.05 with phosphoric acid (~1440 g/L) TS or potassium hydroxide (~110 g/L) TS and then sterilize the solution for 20 minutes in an autoclave at 120 °C.
Phosphate buffer, sterile,1 pH 7.0, TS
Procedure. Dissolve 5.76 g of anhydrous disodium hydrogen phosphate R and 3.55 g of potassium dihydrogen phosphate R in sufficient water to produce 1000 mL. If necessary adjust the pH to 6.95–7.05 with phosphoric acid (~1440 g/L) TS or potassium hydroxide (~110 g/L) TS and then sterilize the solution for 20 minutes in an autoclave at 120 °C.
Phosphate buffer, sterile,1 pH 7.2, TS
Procedure. Dissolve 6.80 g of potassium dihydrogen phosphate R and 1.4 g of sodium hydroxide R in sufficient water to produce 1000 mL. If necessary adjust the pH to 7.1–7.3 with phosphoric acid (~1440 g/L) TS or potassium hydroxide (~110 g/L) TS and then sterilize the solution for 20 minutes in an autoclave at 120 °C.
Phosphate buffer, sterile,1 pH 7.8, TS
Procedure. Dissolve 6.8 g of potassium dihydrogen phosphate R in water, add 45.2 mL of sodium hydroxide (1 mol/L) VS and sufficient water to produce 1000 mL. If necessary adjust the pH to 7.8 with phosphoric acid (~1440 g/L) TS or potassium hydroxide (~110 g/L) TS and then sterilize the solution for 20 minutes in an autoclave at 120 °C.
Phosphate buffer, sterile,1 pH 8.0, TS1
Procedure. Dissolve 16.73 g of dipotassium hydrogen phosphate R and 0.52 g of potassium dihydrogen phosphate R in sufficient water to produce 1000 mL. If necessary adjust the pH to 7.9–8.1 with phosphoric acid (~1440 g/L) TS or potassium hydroxide (~110 g/L) TS and then sterilize the solution for 20 minutes in an autoclave at 120 °C.
Phosphate buffer, sterile,1 pH 8.0, TS2
Procedure. Dissolve 8.95 g of anhydrous disodium hydrogen phosphate R and 0.50 g of potassium dihydrogen phosphate R in sufficient water to produce 1000 mL. If necessary adjust the pH to 7.9–8.1 with phosphoric acid (~1440 g/L) TS or potassium hydroxide (~110 g/L) TS and then sterilize the solution for 20 minutes in an autoclave at 120 °C.
1 The adjustment of the pH, if necessary, should be effected before sterilization of the solution.
Phosphate standard (5 μg/mL) TS
Procedure. Dissolve 0.716 g of potassium dihydrogen phosphate R in sufficient water to produce 1000 mL. Immediately before use dilute 1 mL to 100 mL with water.
Phosphate standard buffer, pH 6.8, TS
Procedure. Dissolve 3.40 g of potassium dihydrogen phosphate R and 3.53 g of anhydrous disodium hydrogen phosphate R in sufficient carbon-dioxide-free water R to produce 1000 mL.
Phosphate standard buffer, pH 7.4, TS
Procedure. Dissolve 1.18 g of potassium dihydrogen phosphate R and 4.30 g of anhydrous disodium hydrogen phosphate R in sufficient carbon-dioxide-free water R to produce 1000 mL.
Phosphate/citrate buffer pH 4.5, TS
Procedure. Dissolve 2.15 g of disodium hydrogen phosphate R in 30 mL of water and adjust the pH of the solution to 4.5 with citric acid (20 g/L) TS.
Phosphate/citrate buffer pH 6.0, TS
Procedure. Dissolve 4.52 g of disodium hydrogen phosphate R in 60 mL of water, add 35 mL of citric acid (20 g/L) TS and if necessary adjust the pH of the solution to 6.0.
Phosphomolybdic acid (80 g/L) TS
A solution of phosphomolybdic acid R containing about 100 g of H3PO4,12MoO3,24H2O per litre.
Phosphomolybdic acid R
H3PO4,12MoO3,24H2O (SRIP, 1963, p. 141).
Phosphomolybdic acid/ethanol TS
Procedure. Dissolve 5 g of phosphomolybdic acid R in sufficient dehydrated ethanol R to produce 100 mL.
Phosphoric acid (~7.8 g/L) TS
Procedure. Dilute 9.2 g of phosphoric acid (~1440 g/L) TS with sufficient water to produce 1000 mL
Phosphoric acid (~105 g/L) TS
Procedure. Mix about 115 g of phosphoric acid (~1440 g/L) TS with 885 g of water.
Phosphoric acid (~1440 g/L) TS
[phosphoric acid R] (SRIP, 1963, p. 141); d ~ 1.7.
Phosphoric acid (~20g/L) TS
Procedure. To 23 g of phosphoric acid (~1440 g/L) TS add 987 g of water and mix.
Phosphoric acid (~2.8 g/L) TS
Procedure. Dilute 2ml of phosphoric acid (~1440 g/L) TS with sufficient water to produce 1000 mL.
Phosphoric acid (~680 g/L) TS
Procedure: Dilute 680 g of phosphoric acid (~1440 g/L) TS with sufficient water to produce 1000 mL.
Phosphorus pentoxide R
P2O5 (SRIP, 1963, p. 142).
Phosphorus, red R
Description. A dark red powder.
Solubility. Insoluble in water and dilute acids.
Soluble matter. Heat 2.0 g with 30 mL of acetic acid (~300 g/L) TS on a water-bath for 15 minutes, cool, dilute to 50 mL, filter, evaporate 25 mL of the filtrate on a water-bath and dry at 110 °C for 2 hours; the residue weighs not more than 50 mg.
Yellow phosphorus. Shake 5.0 g with 20 mL of carbon disulfide R in a glass-stoppered cylinder, filter and immerse in the filtrate a strip of filter-paper, 10 cm by 0.5 cm, previously immersed in copper(II) sulfate (80 g/L) TS, and allow to dry in the air; no stain is produced.
Loss on drying. Dry to constant weight over sulfuric acid (~1760 g/L) TS; it loses not more than 10 mg/g.
Phosphotungstic acid TS
Procedure. Dissolve 25 g of sodium tungstate R in 175 mL of water and add 18.75 mL of phosphoric acid (~1440 g/L) TS. Heat under a reflux condenser for 6 hours, filter and add sufficient water to produce 250 mL.
Storage. Store at a temperature between 2 and 8 °C, protected from light.
Phthalate buffer, pH 3.4, TS
Procedure. Dissolve 2.04 g of potassium hydrogen phthalate R in 50 mL of carbon-dioxide-free water R, add 10.40 mL of hydrochloric acid (0.2 mol/L) VS and dilute with sufficient carbon-dioxide-free water R to produce 200 mL.
Phthalate buffer, pH 3.5, TS
Procedure. Dissolve 2.04 g of potassium hydrogen phthalate R in 50 mL of carbon-dioxide-free water R, add 8.40 mL of hydrochloric acid (0.2 mol/L) VS and dilute with sufficient carbon-dioxide-free water R to produce 200 mL.
Phthalate buffer, pH 4.0, TS
Procedure. Dissolve 2.042 g of potassium hydrogen phthalate R in 50 mL of water, add 0.40 mL of sodium hydroxide (0.2 mol/L) VS and dilute to 200 mL with water.
Phthalein purple R
C32H32N2O12. x H2O. 2,2',2'',2'''-{3-oxo-2-benzofuran-1(3H )-ylidenebis[(6-hydroxyy-5-methyl-3,1-phenylene)methylenenitrilo]}tetraacetic acid.
Description. Yellowish-white to brownish powder.
Solubility. Practically insoluble in water , soluble in ethanol (~750 g/L) TS.
Test for sensitivity. Dissolve 10 mg in 1 mL of ammonia (~260 g/L) TS and dilute to 100 mL with water R. To 5 mL of the solution add 95 mL of water R, 4 mL of ammonia (~260 g/L) TS, 50 mL of ethanol (~750 g/L) TS and 0.1 mL of barium chloride (0.1 mol/L) VS. The solution is blue-violet. Add 0.15 mL of disodium edetate (0.1 mol/L) VS. The solution becomes colourless.
Phthalic anhydride R
C8H4O3.
Description. White lustrous needles.
Solubility. Slightly soluble in water, more soluble in hot water; soluble in ethanol (~750 g/L) TS and ether R.
Melting temperature. About 130 °C.
Phthalic anhydride/pyridine TS
Procedure. Add 42 g of phthalic anhydride R, accurately weighed, to 300 mL of freshly distilled pyridine R (refluxed with barium oxide R) containing less than 1 mg/mL of water in a glass-stoppered 1000 mL flask. Use a dark flask or otherwise prevent exposure to light. Shake vigorously until complete solution is effected and allow to stand overnight for completion of the reaction.
Note: Phthalic anhydride/pyridine TS must be freshly prepared.
Piperazine hydrate R
Piperazine hexahydrate; C4H10N2,6H2O.
Description. Colourless, glossy, deliquescent crystals.
Melting point. 44 °C.
Piperidine R
C5H11N.
Description. A colourless to yellowish liquid; odour, characteristic.
Miscibility. Miscible with water and ethanol (~750 g/L) TS.
Mass density. ρ20 = about 0.86 kg/L.
Refractive index. 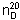 = about 1.454.
= about 1.454.
Boiling temperature. About 106 °C.
Congealing temperature. Between 12 and 15 °C.
Plasma substrate R
Note: Use water-repellent equipment (made from materials such as suitable plastics or suitably silicone-treated glass) for taking and handling blood.
Procedure. Collect a sufficient volume of blood from each of at least 5 sheep. A 285 mL volume of blood collected into 15 mL of anticoagulant solution is recommended but smaller volumes may be collected. The blood should be taken either from a live animal or at the time of slaughter using a needle attached to a cannula that is long enough to reach the bottom of the collecting flask. Discard the first few mL and collect only free-flowing blood. Collect the blood in a sufficient quantity of an anticoagulant solution containing 8.7 g of sodium citrate R and 4 mg of aprotinin R in 100 mL of water to give a final ratio of blood to anticoagulant solution of 19 to 1. During and immediately after collection swirl the flask gently to ensure mixing but do not allow frothing to occur. When collection is complete close the flasks and cool to a temperature between 10 and 15 °C. Then pool the contents of all the flasks, with the exception of any that shows obvious haemolysis or clots, and keep the pooled blood at 10–15°C. Within 4 hours of collection centrifuge the pooled and cooled blood at a speed of 1000–2000 g1 for 30 minutes. Separate the supernatant liquid and centrifuge again at 5000 g for 30 minutes (note: faster centrifugation (20 000 g) may be necessary to clarify the plasma but filtration procedures should not be used). Separate the supernatant liquid and immediately mix thoroughly and distribute the plasma substrate R into small, stoppered containers in portions sufficient for a complete heparin assay (10–30 mL). Without delay, rapidly cool to a temperature below -70 °C by immersing the containers in liquid nitrogen and store at a temperature below -30 °C. The plasma is suitable for use as plasma substrate R in the assay for heparin if, under the conditions of the assay, it gives a clotting time appropriate to the method of detection used and if it provides reproducible, steep, log dose-response curves. Just before use thaw the quantity of plasma substrate R required in a water-bath at 37 °C and gently swirl until thawing is complete; once thawed it should be kept between 10 and 20 °C and used without delay. The thawed plasma substrate R may be slightly centrifuged if necessary but do not use any filtration procedures.
1 Acceleration due to gravity = 9.81m/s2.
Platinic chloride (60 g/L) TS
A solution of platinic chloride R containing about 63 g of H2PtCl6, per litre.
Platinic chloride R
H2PtCl6,6H2O (SRIP, 1963, p. 144).
Poly(dimethyl)(diphenyl)siloxane R
Stationary phase for gas chromatography. Contains 95% of methyl groups and 5% of phenyl groups.
Poly(dimethyl)siloxane R
Stationary phase for gas chromatography. Contains polysiloxane substituted with 100% of methyl groups
Polydimethylsiloxane R
A commercially available reagent of suitable grade for use in gas chromatography.
Polyethyleneglycol, base-deactivated R
Cross-linked, base-deactivated polyethyleneglycol, specially designed to be used as a stationary phase for gas chromatographic analysis of amine.
Polyoxyethylated castor oil R
Light yellow liquid. It becomes clear above 26 °C.
Polyoxyethylene (23) lauryl ether R.
Synonym: Brij 35, C58H120O24;1199.6, CAS 9002-92-0.
A commercially available reagent of suitable grade.
Polysorbate 80 R
The mono ester of oleic acid and tripolyethyleneglycol 300-sorbitan ether.
Description. Lemon to amber coloured, oily liquid.
Miscibility. Miscible with water, producing an odourless and nearly colourless solution. Miscible with ethanol (~750 g/L) TS, ethyl acetate R and vegetable oils; immiscible with mineral oils.
Potassio-cupric tartrate TS
Procedure. Dissolve 7 g of copper(II) sulfate R in sufficient water to produce 100 mL. Separately dissolve 35 g of potassium sodium tartrate R and 10 g of sodium hydroxide R in 100 mL of water. Shortly before use, mix together equal volumes of both solutions.
Potassio-mercuric iodide TS
Procedure. Dissolve 1.355 g of mercuric chloride R in 60 mL of water; separately dissolve 5 g of potassium iodide R in 20 mL of water; mix the two solutions and dilute to 100 mL with water.
Potassio-mercuric iodide, alkaline, TS
Procedure. Dissolve 3.5 g of potassium iodide R and 1.25 g of mercuric chloride R in 80 mL of water, add while stirring a cold, saturated solution of mercuric chloride R in water until a slight red precipitate remains. Then add 12 g of sodium hydroxide R and mix to dissolve, add a little more of the saturated solution of mercuric chloride R and sufficient water to produce 100 mL; allow to stand for 24 hours and decant the clear liquid.
Potassium acetate R
C2H3KO2 (SRIP, 1963, p. 144).
Potassium acetate TS
Procedure. Dissolve 100 g of potassium acetate R in sufficient glacial acetic acid R to produce 1000 mL.
Potassium antimonate R
KSbO3 (SRIP, 1963, p. 145).
Potassium antimonate TS
Procedure. Boil 2 g of potassium antimonate R with 95 mL of water until it has dissolved. Cool rapidly and add 50 mL of potassium hydroxide (1 mol/L) VS and 5 mL of sodium hydroxide (1 mol/L) VS. Allow to stand for 24 hours and dilute with sufficient water to produce 150 mL.
Sensitivity to sodium. To 10 mL add 7 mL of sodium hydroxide (0.1 mol/L) VS; a white, crystalline precipitate is formed within 15 minutes.
Note. Potassium antimonate TS must be freshly prepared.
Potassium bicarbonate R
KHCO3 (SRIP, 1963, p. 145).
Potassium bromate (0.00833 mol/L) VS
Potassium bromate R dissolved in water to contain 1.392 g of KBrO3 in 1000 mL.
Potassium bromate (0.0167 mol/L) VS
Potassium bromate R dissolved in water to contain 2.784 g of KBrO3 in 1000 mL.
Potassium bromate (0.0333 mol/L) VS
Potassium bromate R dissolved in water to contain 5.562 g of KBrO3 in 1000 mL.
Potassium bromate (50 g/L) TS
A solution of potassium bromate R containing about 50 g of KBrO3 per litre.
Potassium bromate R
KBrO3 (SRIP, 1963, p. 147).
Potassium bromide (0.119 g/L) TS
A solution of potassium bromide R containing about 0.1190 g of KBr per litre.
Potassium bromide (100 g/L) TS
A solution of potassium bromide R containing about 100 g of KBr per litre.
Potassium bromide (125 g/L) TS
A solution of potassium bromide R containing about 125 g of KBr per litre.
Potassium bromide IR
Potassium bromide R that complies with the following test: the infrared absorption spectrum of a disc prepared as described in Method 3 under 1.7 Spectrophotometry in the infrared region from potassium bromide R, previously dried at 250 °C for 1 hour, has a substantially flat baseline over the range 4000–670 cm-1; it exhibits no maxima with an absorbance greater than 0.1 above the baseline with the exception of maxima due to water at 3440 and 1630 cm-1.
Potassium bromide R
KBr (SRIP, 1963, p. 148).
Potassium carbonate R
K2CO3, 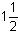 H2O.
H2O.
Description. Small granular crystals.
Solubility. Very soluble in water; practically insoluble in ethanol (~750 g/L) TS.
Potassium carbonate, anhydrous, R
K2CO3.
Description. Granules or a granular powder; hygroscopic.
Solubility. Soluble in 1 part of water; practically insoluble in ethanol (~750 g/L) TS.
Potassium carbonate (150 g/L) TS
Procedure. Dissolve 15 g of anhydrous potassium carbonate R in 100 mL of water R.
Potassium chloride (100 g/L) TS
A solution of potassium chloride R containing about 100 g of KCl per litre.
Potassium chloride (350 g/L) TS
A saturated solution of potassium chloride R containing about 350 g/L of KCl.
Potassium chloride IR
Potassium chloride R that complies with the following test: the infrared absorption spectrum of a disc prepared as described in Method 3 under 1.7 Spectrophotometry in the infrared region, from potassium chloride R, previously dried at 250 °C for 1 hour, has a substantially flat baseline over the range 4000–670 cm-1; it exhibits no maxima with an absorbance greater than 0.1 above the baseline, with the exception of maxima due to water at 3440 and 1630 cm-1.
Potassium chloride R
KCl (SRIP, 1963, p. 151).
Potassium chromate (100 g/l) TS
A solution of potassium chromate R containing about 97 g of K2CrO4 per litre (approximately 0.5 mol/L).
Potassium chromate R
K2CrO4 (SRIP, 1963, p. 152).
Potassium cyanide (100 g/L) TS
A solution of potassium cyanide R containing about 100 g of KCN per litre.
Potassium cyanide (50 g/L) TS
A solution of potassium cyanide R containing about 50 g of KCN per litre.
Potassium cyanide PbTS.
Procedure. Dissolve 10 g of potassium cyanide R in 90 mL of water, add 2 mL of hydrogen peroxide (~60 g/L) TS, allow to stand for 24 hours, dilute with water to 100 mL and filter.
Potassium cyanide R
KCN (SRIP, 1963, p. 153).
Potassium dichromate (0.0167 mol/L) VS
Potassium dichromate R dissolved in water to contain 4.904 g of K2Cr2O7 in 1000 mL.
Potassium dichromate (100 g/L) TS
A solution of potassium dichromate R containing about 98 g of K2Cr2O7 per litre (approximately 0.4 mol/L).
Potassium dichromate R
K2Cr2O7 (SRIP, 1963, p. 154).
Potassium dichromate R1
Potassium dichromate R containing not less than 99.9% of K2Cr2O7.
Potassium dichromate TS
Procedure. Dissolve about 60 mg, accurately weighed and previously dried at 130 °C, of potassium dichromate R1 in sufficient sulfuric acid (0.005 mol/L) VS to produce 1000.0 mL.
Potassium dichromate TS2
Procedure. Dissolve 1 g of potassium dichromate R in 60 mL of water and cautiously add 7.5 mL of sulfuric acid (~1760 g/L) TS.
Potassium dichromate TS3
Procedure. Dissolve 0.5 g of potassium dichromate R in sufficient sulfuric acid (~100 g/L) TS to produce 100 mL.
Potassium dihydrogen phosphate (6.8 g/L) TS
A solution of potassium dihydrogen phosphate R containing 6.8 g of KH2PO4 per litre (0.05 mol/L).
Potassium dihydrogen phosphate (13.6 g/L) TS
A solution of potassium dihydrogen phosphate R containing 13.6 g of KH2PO4 per litre (0.1 mol/L).
Potassium dihydrogen phosphate (27.2 g/L) TS
A solution of potassium dihydrogen phosphate R containing 27.2 g of KH2PO4 per litre (0.2 mol/L).
Potassium dihydrogen phosphate (70 g/L) TS
A solution of potassium dihydrogen phosphate R containing about 70 g of KH2PO4 per litre.
Potassium dihydrogen phosphate (100 g/L) TS.
A solution of potassium dihydrogen phosphate R containing about 100 g of KH2PO4 per litre.
Potassium dihydrogen phosphate R
KH2PO4 (SPRI, 1963, p. 155).
Potassium ferricyanide (10 g/L) TS
Procedure. Wash about 1 g of crystalline potassium ferricyanide R with a little water and dissolve the washed crystals in sufficient water to produce 100 mL.
Note: Potassium ferricyanide (10 g/L) TS must be freshly prepared.
Potassium ferricyanide (50 g/L) TS
Procedure. Wash about 5 g of crystalline potassium ferricyanide R with a little water and dissolve the washed crystals in sufficient water to produce 100 mL.
Note: Potassium ferricyanide (50 g/L) TS must be freshly prepared.
Potassium ferrocyanide (~53 g/L) TS
A solution of potassium ferrocyanide R containing about 53 g of K4Fe(CN)6 per litre.
Potassium ferricyanide R
K3Fe(CN)6 (SRIP, 1963, p. 156).
Potassium ferrocyanide (45 g/L) TS
A solution of potassium ferrocyanide R containing about 50 g of K4Fe(CN)6 per litre.
Potassium ferrocyanide R
K4Fe(CN)6,3H2O (SRIP, 1963, p. 156).
Potassium gluconate R
C6H11KO7
Potassium hydrogen phthalate R
C8H5KO4 (SRIP, 1963, p. 157).
Potassium hydrogen phthalate standard TS
Procedure. Dissolve 10.21 g of potassium hydrogen phthalate R, previously dried at 120 °C, in sufficient carbon-dioxide-free water R to produce 1000 mL. The pH of this solution is defined as having the value of 4.000 at 15 °C.
Potassium hydrogen sulfate R
KHSO4 (SRIP, 1963, p. 146).
Potassium hydrogen tartrate R
C4H5KO6 (SRIP, 1963, p. 158).
Potassium hydrogen tartrate standard TS
Procedure. Add 2 g of potassium hydrogen tartrate R to 100 mL of carbon-dioxide-free water R contained in a glass-stoppered flask and shake the flask vigorously. Let the temperature of the solution reach room temperature, allow the solid to settle and remove it by filtration or decantation.
Note: Potassium hydrogen tartrate standard TS must be freshly prepared.
Potassium hydroxide (~55 g/L) TS
A solution of potassium hydroxide R containing about 55 g/L of KOH (approximately 1 mol/L).
Potassium hydroxide (~110 g/L) TS
A solution of potassium hydroxide R containing about 112 g/L of KOH (approximately 2 mol/L).
Potassium hydroxide (~400 g/L) TS
A solution of potassium hydroxide R containing about 400 g of KOH per litre.
Potassium hydroxide (~450 g/L) TS
A solution of potassium hydroxide R containing about 450 g of KOH per litre.
Potassium hydroxide (~560 g/L) TS
A solution of potassium hydroxide R containing about 560 g of KOH per litre.
Potassium hydroxide (0.01 mol/L) VS
Potassium hydroxide R dissolved in water to contain 0.5610 g of KOH in 1000 mL.
Method of standardization. Ascertain the exact concentration of the solution following the method described under potassium hydroxide (1 mol/L) VS.
Potassium hydroxide (0.1 mol/L) VS
Potassium hydroxide R dissolved in water to contain 5.610 g of KOH in 1000 mL.
Method of standardization. Ascertain the exact concentration of the solution following the method described under potassium hydroxide (1 mol/L) VS.
Potassium hydroxide/ethanol (0.1 mol/L) VS
Potassium hydroxide R dissolved in ethanol (~710 g/L) TS to contain 5.61 g of KOH in 1000 mL.
Method of standardization. Ascertain the exact concentration of the solution following the method described under potassium hydroxide/ethanol (0.5 mol/L) VS
Potassium hydroxide (0.5 mol/L) VS
Potassium hydroxide R dissolved in water to contain 28.05 g of KOH in 1000 mL.
Method of standardization. Ascertain the exact concentration of the solution following the method described under potassium hydroxide (1 mol/L) VS.
Potassium hydroxide (1 mol/L) VS
Potassium hydroxide R dissolved in water to contain 56.10 g of KOH in 1000 mL.
Method of standardization. Ascertain the exact concentration of the 1 mol/L solution in the following manner: dry about 5 g of potassium hydrogen phthalate R at 105 °C for 3 hours and weigh accurately. If the potassium hydrogen phthalate is in the form of large crystals they should be crushed before drying. Dissolve in 75 mL of carbon-dioxide-free water R and titrate with the potassium hydroxide solution using phenolphthalein/ethanol TS as indicator. Each 0.2042 g of potassium hydrogen phthalate is equivalent to 1 mL of potassium hydroxide (1 mol/L) VS. Standard solutions of potassium hydroxide should be restandardized frequently.
Storage. Solutions of alkali hydroxides absorb carbon dioxide when exposed to air. They should therefore be stored in bottles with suitable non-glass, well-fitting stoppers, provided with a tube filled with soda lime R.
Potassium hydroxide R
KOH (SRIP, 1963, p. 159).
Potassium hydroxide/ethanol (0.02 mol/L) VS
Potassium hydroxide R dissolved in ethanol (~710 g/L) TS to contain 1.122 g of KOH in 1000 mL.
Method of standardization. Ascertain the exact concentration of the solution following the method described under potassium hydroxide/ethanol (0.5 mol/L) VS.
Potassium hydroxide/ethanol (0.5 mol/L) VS
Potassium hydroxide R dissolved in ethanol (~710 g/L) TS to contain 28.05 g of KOH in 1000 mL.
Method of standardization. Ascertain the exact concentration of the 0.5 mol/L solution in the following manner: dilute 25.0 mL of hydrochloric acid (0.5 mol/L) VS with 50 mL of water and titrate with the potassium hydroxide/ethanol solution using phenolphthalein/ethanol TS as indicator.
Potassium hydroxide/ethanol (1 mol/L) VS
Potassium hydroxide R dissolved in ethanol (~710 g/L) TS to contain 56.10 g of KOH in 1000 mL.
Method of standardization. Ascertain the exact concentration of the solution following the method described under potassium hydroxide/ethanol (0.5 mol/L) VS.
Potassium hydroxide/ethanol TS1
Procedure. Dissolve 40 g of potassium hydroxide R in 20 mL of water and add sufficient ethanol (~750 g/L) TS to produce 1000 mL. Allow to stand overnight and pour off the clear liquid.
Potassium hydroxide/ethanol TS2
Procedure. Dissolve 112 g of potassium hydroxide R in sufficient ethanol (~710 g/L) TS to produce 1000 mL (approximately 2 mol/L).
Potassium hydroxide/methanol TS
Procedure. Dissolve 30 g of potassium hydroxide R in sufficient methanol R to produce 1000 mL.
Potassium iodate (0.01 mol/L) VS
Potassium iodate R dissolved in water to contain 2.140 g of KIO3 in 1000 mL.
Method of standardization. Ascertain the exact concentration of the solution following the method described under potassium iodate (0.05 mol/L) VS.
Potassium iodate (0.05 mol/L) VS
Potassium iodate R dissolved in water to contain 10.70 g of KIO3 in 1000 mL.
Method of standardization. Ascertain the exact concentration of the 0.05 mol/L solution in the following manner. Place 10.0 mL of the potassium iodate solution in a glass-stoppered flask, dilute with 200 mL of water, add 2 g of potassium iodide R and 25 mL of sulfuric acid (~100 g/L) TS. Allow the solution to stand for 10 minutes and titrate the liberated iodine with sodium thiosulfate (0.1 mol/L) VS, adding 3 mL of starch TS as the end-point is approached. Correct for a blank determined on the same quantities of the same reagents.
Potassium iodate (3.6 mg/L) TS
A freshly prepared solution of potassium iodate R containing 3.6 mg of KIO3 per litre.
Potassium iodate R
KIO3 (SRIP, 1963, p. 160).
Potassium iodide (100 g/L) TS
A solution of potassium iodide R containing about 100 g of KI per litre.
Potassium iodide (160g/L) TS
A solution of potassium iodide R containing about 160 g of KI per litre.
Potassium iodide (300 g/L) TS
A solution of potassium iodide R containing about 300 g of KI per litre.
Potassium iodide (400 g/L) TS
A solution of potassium iodide R containing about 400 g of KI per litre.
Potassium iodide (60 g/L) TS
A solution of potassium iodide R containing about 60 g of KI per litre.
Potassium iodide (80 g/L) TS
A solution of potassium iodide R containing about 83 g/L of KI (approximately 0.5 mol/L).
Potassium iodide AsR
Potassium iodide R that complies with the following test: Dissolve 10 g of potassium iodide R in 25 mL of hydrochloric acid (~250 g/L) AsTS and 35 mL of water, add 2 drops of stannous chloride AsTS and apply the general test for arsenic; no visible stain is produced.
Potassium iodide R
KI (SRIP, 1963, p. 161).
Potassium iodide, iodinated TS
Procedure. Dissolve 500 mg of iodine R and 1.5 g of potassium iodide R in water R and dilute to 25 mL with the same solvent.
Potassium iodide/starch TS1
Procedure. Dissolve 10 g of potassium iodide R in 95 mL of water and add to it 5 mL of starch TS.
Note: Potassium iodide/starch TS1 must be freshly prepared.
Potassium iodobismuthate TS1
Procedure. Dissolve 100 g of tartaric acid R in 400 mL of water and add 8.5 g of bismuth oxynitrate R. Shake the solution for 1 hour, add 200 mL of potassium iodide (400 g/L) TS and mix. Allow to stand for 24 hours and filter.
Potassium iodobismuthate TS2
Procedure. Dissolve 100 g of tartaric acid R in 500 mL of water and add 50 mL of potassium iodobismuthate TS1.
Potassium iodobismuthate TS3
Stock solution. Suspend 1.7 g of bismuth subnitrate R and 20 g of tartaric acid R in 40 mL of water R. To the suspension add 40 mL of potassium iodide (400 g/L) TS and stir for 1 hour. Filter. The solution may be kept for several days in an amber glass bottle.
Spray solution. Prepare immediately before use. Mix 1 volume of the stock solution with 3 volumes of water R.
Potassium iodobismuthate/acetic acid TS
Procedure. Dissolve 8 g of potassium iodide R in 20 mL of water and add to it a solution composed of 0.85 g of bismuth oxynitrate R dissolved in 40 mL of water and 10 mL of glacial acetic acid R.
Potassium iodoplatinate TS
Procedure. Dissolve 2.5 g of platinic chloride R in 50 mL of water, add 45 mL of a 0.1 g/mL solution of potassium iodide R and dilute to 100 mL with water.
Storage. Store in amber glass containers.
Potassium iodoplatinate TS2
Procedure. Dissolve 0.25 g of platinic chloride R in 2.5 mL of water, add 45 mL of potassium iodide (100 g/L) TS and dilute with sufficient acetone R to produce 100 mL.
Potassium nitrate R
KNO3 (SRIP, 1963, p. 162).
Potassium nitrite (100 g/L) TS
A solution of potassium nitrite R containing about 100 g/L of KNO2.
Potassium nitrite R
KNO2.
Description. White or slightly yellow, deliquescent granules or rods.
Solubility. Soluble in 0.35 part of water; slightly soluble in ethanol (~750 g/L) TS.
Potassium periodate R
KIO4 (SRIP, 1963, p. 164).
Potassium periodate TS
Procedure. To 2.8 g of potassium periodate R add 200 mL of water followed by 20 mL of sulfuric acid (~1760 g/L) TS added drop by drop while shaking to effect solution; cool and add sufficient water to produce 1000 mL.
Potassium permanganate (0.0002 mol/L) VS
Potassium permanganate R dissolved in water to contain 31.61 mg of KMnO4 in 1000 mL.
Method of standardization. Ascertain the exact concentration of the solution following the method described under "Potassium permanganate (0.02 mol/L) VS".
Potassium permanganate (0.002 mol/L) VS
Potassium permanganate R dissolved in water to contain 0.3161 g of KMnO4 in 1000 mL.
Method of standardization. Ascertain the exact concentration of the solution following the method described under potassium permanganate (0.02 mol/L) VS.
Potassium permanganate (0.02 mol/L) VS
Potassium permanganate R dissolved in water to contain 3.161 g of KMnO4 in 1000 mL.
Method of standardization. Ascertain the exact concentration of the 0.02 mol/L solution in the following manner: dissolve about 0.2 g, accurately weighed, of sodium oxalate R, previously dried to constant weight at 110 °C, in 250 mL of water. Add 7 mL of sulfuric acid (~1760 g/L) TS, heat to about 70 °C and then slowly add the permanganate solution from a burette, with constant stirring, until a pale pink colour, which persists for 15 seconds, is produced. The temperature at the conclusion of the titration should be not less than 60 °C. Every 6.7 mg of sodium oxalate are equivalent to 1 mL of potassium permanganate (0.02 mol/L) VS. Potassium permanganate solutions should be restandardized frequently.
Storage. Store the solution in tightly closed containers, protected from light.
Potassium permanganate (~ 0.3 g/L) TS
A solution of potassium permanganate R containing about 0.3 g of KMnO4 per litre.
Potassium permanganate (~25 g/L) TS
A solution of potassium permanganate R containing about 25 g of KMnO4 per litre.
Potassium permanganate (~10 g/L) TS
A solution of potassium permanganate R containing about 10 g/L of KMnO4.
Potassium permanganate (~1g/L) TS
A solution of potassium permanganate R containing about 1 g of KMnO4 per litre.
Potassium permanganate R
KMnO4 (SRIP, 1963, p. 165).
Potassium permanganate, basic (~5 g/L) TS
A solution of potassium permanganate R containing about 5 g of KMnO4 per litre of sodium hydroxide (~40 g/L) TS.
Potassium permanganate, basic (~1 g/L) TS
A solution of potassium permanganate R containing about 1 g of KMnO4 per litre of sodium hydroxide (~40 g/L) TS.
Potassium permanganate/phosphoric acid TS
Procedure. Dissolve 3 g of potassium permanganate R in a mixture of 15 mL of phosphoric acid (~1440 g/L) TS and 70 mL of water and dilute to 100 mL with water.
Potassium pyroantimonate R
KSb(OH)6
Description. White or almost white, crystals or crystalline powder.
Solubility. Sparingly soluble in water R.
Potassium pyroantimonate (13 g/L) TS
Dissolve 1.95 g of potassium pyroantimonate R in 95 mL of hot water R. Cool quickly and add a solution containing 2.5 g of potassium hydroxide R in 50 mL of water R and 1 mL of dilute sodium hydroxide (~85 g/L) TS. Allow to stand for 24 hours, filter and dilute to 150 mL with water R.
Potassium sodium tartrate R
[sodium potassium tartrate R] C4H4KNaO6,4H2O (SRIP, 1963, p. 193).
Potassium sulfate (0.1 g/L) TS
A solution of potassium sulfate R containing about 0.1 g of K2SO4 per litre.
Potassium sulfate (174 mg/L) TS
Procedure. Dissolve 174 mg, accurately weighed, of potassium sulfate R in sufficient water to produce 1000 mL.
Potassium sulfate R
K2SO4 (SRIP, 1963, p. 165).
Potassium tetraoxalate R
C4H3KO8,2H2O (SRIP, 1963, p. 166).
Potassium tetraoxalate standard TS
Procedure. Dissolve 25.42 g of potassium tetraoxalate R in sufficient carbon-dioxide-free water R to produce 1000 mL.
Potassium thiocyanate (200 g/L) TS
A solution of potassium thiocyanate R containing 200 g of KCNS per litre.
Potassium thiocyanate R
KCNS. Contains not less than 99.0% of KCNS, calculated with reference to the dried substance.
Description. Colourless crystals.
Solubility. Soluble in 0.5 part of water and in 15 parts of dehydrated ethanol R.
Alkalinity. A 0.1 g/mL solution in carbon-dioxide-free water R is not alkaline to bromothymol blue/ethanol TS.
Ammonia. Boil 1.0 g with 5 mL of sodium hydroxide (~80 g/L) TS; no ammonia is evolved.
Chlorides. Dissolve 1.0 g in a solution of 1 g of ammonium nitrate R in 30 mL of hydrogen peroxide (~60 g/L) TS containing not more than 1 μg/g of chlorides, add 1 g of sodium hydroxide R and warm gently; when the vigorous reaction subsides add a further 30 mL of hydrogen peroxide (~60 g/L) TS and boil for 2 minutes. Cool, add 5 mL of nitric acid (~1000 g/L) TS and 1 mL of silver nitrate (40 g/L) TS; any opalescence produced is not greater than that produced by treating 1 mL of hydrochloric acid (0.01 mol/L) VS in the same manner.
Sulfates. Dissolve 0.50 g in 20 mL of water and proceed as described under 2.2.2 Limit test for sulfates; not more than 1.0 mg/g.
Other sulfur compounds. Dissolve 1.0 g in 50 mL of water, add 2 mL of hydrochloric acid (~70 g/L) TS, and titrate with iodine (0.05 mol/L) VS; not more than 0.5 mL of iodine (0.05 mol/L) VS is required.
Loss on drying. Dry to constant weight at 105 °C; it loses not more than 20 mg/g.
Assay. Dissolve about 0.4 g, accurately weighed, in 50 mL of water, add 5 mL of nitric acid (~1000 g/L) TS, 50 mL of silver nitrate (0.1 mol/L) VS and 5 mL of ferric ammonium sulfate (45 g/L) TS and titrate the excess of silver nitrate with ammonium thiocyanate (0.1 mol/L) VS. Each mL of silver nitrate (0.1 mol/L) VS is equivalent to 9.718 mg of KCNS.
Note: Potassium thiocyanate R is deliquescent.
1,3 Propanediol R
C3H8O2; CAS Reg. No. 504-63-2.
Contains not less than 99.0% of C3H8O2.
2-Propanol R
[iso-propanol R]; isopropyl alcohol; C3H8O (SRIP, 1963, p. 167).
1-Propanol R
n- Propanol; propan-1-ol, C3H8O.
Description. A clear, colourless liquid.
Miscibility. Miscible with water and ethanol (~750 g/L) TS.
Boiling range. Not less than 95% distils between 95 and 98 °C.
Mass density. ρ20 = about 0.803 kg/L.
Residue on evaporation. Evaporate on a water-bath and dry to constant weight at 105 °C; it leaves a residue of not more than 0.1 mg/g.
Propylene glycol R.
C3H8O2 (SRIP, 1963, p. 168).
Pyridine R
C5H5N (SRIP, 1963, p. 169).
Pyridine, anhydrous, R
Pyridine R that has been dried by allowing it to stand over sodium hydroxide R.
Pyridine/acetic anhydride TS
Procedure. Mix 3 volumes of freshly distilled pyridine R with 1 volume of freshly distilled acetic anhydride R.
Note: Pyridine/acetic anhydride TS must be freshly prepared.
Pyrogallol R
Pyrogallic acid; 1,2,3-trihydroxybenzene, C6H6O3 (SRIP, 1963, p. 170).
Pyrogallol, alkaline, TS
Procedure. Dissolve 0.5 g of pyrogallol R in 2 mL of water. Dissolve separately 12 g of potassium hydroxide R in 8 mL of water. Immediately before use mix the two solutions.