Reagents, test solutions and volumetric solutions: T - Tannic acid (50 g/L) TS.... Tyrosine R
Tannic acid (50 g/L) TS
A solution of tannic acid R containing about 50 g of C76H52O46 per litre.
Tannic acid R
C76H52O46 (SRIP, 1963, p. 205).
Tartaric acid (10 g/L) TS
A solution of tartaric acid R containing about 10 g of C4H6O6 per litre.
Tartaric acid (200 g/L) TS
A solution of tartaric acid R containing about 200 g of C4H6O6 per litre.
Tartaric acid (5 g/L) TS
A solution of tartaric acid R containing about 5 g of C4H6O6 per litre.
Tartaric acid R
C4H6O6 (SRIP, 1963, p. 205).
p-Terphenyl R
1,4-Diphenylbenzene, C18H14. Suitable for scintillation counting.
Thioglycolic acid R. See Mercaptoacetic acid R.
Testosterone propionate R
C22H32O3. Use testosterone propionate as described in the monograph for Testosterone propionate.
Testosterone propionate/ethanol TS
Procedure. Dissolve 10 mg of testosterone propionate R in sufficient ethanol (~750 g/L) TS to produce 10 mLl.
Tetrabromophenolphthalein ethyl ester R
3',3",5',5"-Tetrabromophenolphthalein, ethyl ester; C22H14Br4O4. Use a suitable reagent grade.
Tetrabromophenolphthalein ethyl ester TS
Procedure. Dissolve 0.10 g of tetrabromophenolphthalein ethyl ester R in sufficient glacial acetic acid R to produce 100 mL.
Note: Tetrabromophenolphthalein ethyl ester TS should be freshly prepared.
Tetrabutylammonium hydrogen sulfate R
TBAHS; C16H37NO4S.
A commercially available reagent of suitable grade.
Description. Colourless crystals or a white, crystalline powder.
Solubility. Freely soluble in water and methanol R; soluble in ethanol (~750 g/L) TS producing a slightly hazy, colourless solution.
Absorbance. A 0.05 g/mL solution of a 1 cm layer measured at a wavelength between 240 nm and 300 nm has an absorbance of not greater than 0.05.
Melting range. 169–173 °C.
Tetrabutylammonium hydroxide (0.1 mol/L) VS
Procedure. Dissolve 40 g of tetrabutylammonium iodide R in 90 mL of dehydrated ethanol R, add 20 g of finely powdered, purified silver oxide R and shake vigorously for 1 hour. Centrifuge a small volume of the mixture and test the supernatant liquid for iodides. If a positive reaction is obtained add an additional 2 g of silver oxide R and shake for a further 30 minutes. Repeat this procedure until the liquid is free from iodides, filter the mixture through a fine sintered glass filter and rinse the reaction vessel and the filter with 3 quantities of dry benzene R, each of 50 mL. Add the washings to the filtrate and dilute to 1000 mL with dry benzene R. Pass dry carbon-dioxide-free nitrogen R through the solution for 5 minutes.
Method of standardization. Titrate 10 mL of dimethylformamide R with the tetrabutylammonium hydroxide solution using 3 drops of thymol blue/methanol TS as indicator until a pure blue colour is obtained. Immediately add about 0.06 g of benzoic acid R, accurately weighed, stir to effect solution and titrate with the tetrabutylammonium hydroxide solution until the full blue colour of the indicator is again obtained. The solution must be protected from atmospheric carbon dioxide throughout the titration. From the volume of the titrant used in the second titration ascertain the exact concentration of the 0.1 mol/L solution. Each 12.21 mg of benzoic acid is equivalent to 1 mL of tetrabutylammonium hydroxide (0.1 mol/L) VS.
Note: Tetrabutylammonium hydroxide (0.1 mol/L) VS must be standardized immediately before use.
Tetrabutylammonium hydroxide TS
C16H37NO. A solution in water containing about 400 g of C16H37NO per litre (~1.5 mol/L).
Tetrabutylammonium hydroxide/methanol TS
Procedure. Dilute a sufficient volume of tetrabutylammonium hydroxide TS with methanol R to obtain a solution containing 0.25 g of C16H37NO per mL.
Tetrabutylammonium iodide R
C16H36IN. Contains not less than 98.0% of C16H36IN.
Description. White or slightly cream-coloured crystals or a crystalline powder.
Solubility. Soluble in ethanol (~750 g/L) TS.
Sulfated ash. Not more than 0.2 mg/g.
Assay. Dissolve about 1.2 g, accurately weighed, in 30 mL of water. Add 50 mL of silver nitrate (0.1 mol/L) VS and 5 mL of nitric acid (~130 g/L) TS. Titrate the excess of silver nitrate with ammonium thiocyanate (0.1 mol/L) VS using ferric ammonium sulfate (45 g/L) TS as indicator. Each mL of silver nitrate (0.1 mol/L) VS is equivalent to 36.94 mg of C16H36IN.
n-Tetradecane R
C14H30.
Description. A clear and colourless liquid.
Miscibility. Miscible with ethanol (~750 g/L) TS.
Mass density. ρ20 = about 0.76 kg/L.
Refractive index. 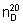 = 1.428–1.429.
= 1.428–1.429.
Tetrachloroethane R
1,1,2,2-Tetrachloroethane, C2H2Cl4.
Description. A clear, colourless liquid.
Miscibility. Miscible with 400 parts of water; miscible with ethanol (~750 g/L) TS and ether R.
Boiling range. Not less than 95% distils between 142 and 147 °C.
Refractive index. ρ20 = 1.493–1.495.
Mass density. 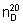 = 1.590 - 1.595 kg/l.
= 1.590 - 1.595 kg/l.
Tetradecylammonium bromide R
C40H84BrN. Chromatographic reagent grade of commerce.
Description. White to almost white crystals, or a crystalline powder.
Melting range. Between 88–89 °C.
Tetraheptylammonium bromide R
C28H60BrN. Chromatographic reagent grade of commerce.
Description. White, flaky powder.
Melting range. Between 89-91 °C.
Tetrahydrofuran R
C4H8O.
Description. A colourless liquid; odour, characteristic, pungent.
Boiling point. About 66 °C.
Mass density. ρ20 = 0.884–0.886 kg/L.
Storage. Store in small, well-filled containers, protected from light.
Labelling. The name and concentration of any suitable preservative, not exceeding 0.1%, should be stated on the label.
Tetramethylammonium hydroxide (~100 g/L) TS
Contains about 100 g/L of (CH3)4NOH in water.
Description. A clear and colourless liquid; odour, strong, ammonia-like.
Residue on evaporation. Evaporate 5 mL on a water-bath and dry at 105 °C for 1 hour; it leaves a residue of not more than 1.0 mg (0.2 mg/g).
Ammonia and other amines. Weigh accurately a quantity of the solution, equivalent to about 0.3 g of (CH3)4NOH, in a low-form weighing bottle tared with 5 mL of water. Add a slight excess of hydrochloric acid (1 mol/L) VS (about 4 mL), evaporate to dryness on a water-bath and dry at 105 °C for 2 hours. The weight of the residue obtained, multiplied by 0.8317, represents the quantity in mg of (CH3)4NOH corresponding to within ± 0.2% of that found in the assay.
Assay. Weigh accurately a glass-stoppered flask containing about 15 mL of water. Add a quantity of the solution equivalent to about 0.2 g of (CH3)4NOH and weigh again. Add methyl red/ethanol TS and titrate with hydrochloric acid (0.1 mol/L) VS. Each mL of hydrochloric acid (0.1 mol/L) VS is equivalent to 9.115 mg of (CH3)4NOH.
Storage. Store in a tightly closed container.
Tetramethylammonium hydroxide/ethanol TS
Procedure. Dilute 10 mL of tetramethylammonium hydroxide (~100 g/L) TS with sufficient ethanol (~750 g/L) TS to produce 100 mL.
N,N,N',N'-Tetramethylethane-1,2-diamine R
C6H16N2
Molecular weight. 116.2.
Other name. N,N,N’,N’ -Tetramethylethylenediamine.
Description. Colourless liquid, miscible with water and with ethanol (~710 g/L) TS.
Relative density![]() . About 0.78.
. About 0.78.
Boiling point. About 121 °C.
Thallium standard (2 µg/mL Tl) TS
Procedure. Transfer 235.0 mg of thallous chloride R accurately weighed, to a 1000 mL volumetric flask, dilute to volume with water R and mix. Transfer 1.0 mL of this solution to a 100 mL volumetric flask, dilute to volume with saline TS containing 0.9% of benzyl alcohol R and mix. Each mL of this solution contains 2 µg of thallium.
Thioacetamide R
C2H5NS = 75.13 (62-55-5).
General reagent grade of commerce.
White crystals or crystalline powder; melting point, about 113 °C.
Thioacetamide reagent TS
Add 1 mL of a mixture of 15 mL of 1m sodium hydroxide, 5 mL of water and 20 mL of glycerol (85%) to 0.2 mL of thioacetamide solution TS, heat in a water bath for 20 seconds, cool and use immediately.
Thioacetamide solution TS
A 4% w/v solution of thioacetamide R.
Thioacetamide, alkaline, TS
Procedure. Dissolve 0.4 g of thioacetamide R in 10 mL of water. Immediately before use add 0.2 mL of this solution to 1 mL of a mixture of 15 mL of sodium hydroxide (1 mol/L) VS, 5 mL of water and 20 mL of glycerol R. Heat on a water-bath for 20 seconds.
Thioglycollic acid R
C2H4O2S 2-Mercaptoacetic acid (MW 92.1) CAS [68-11-1]
A colourless liquid, soluble in alcohol and miscible with water.
Thiourea (0.1 g/L) TS
A solution of thiourea R containing 0.1 g of CH4N2S per litre.
Thiourea R
CH4N2S (SRIP, 1963, p. 207).
Thorin (2 g/L) TS
Procedure. Dissolve 0.2 g of thorin R in sufficient water to produce 100 mL.
Storage. Store the solution protected from light.
Shelf-life. Use within 1 week of preparation.
Thorin R
2,7-Disodium 4-[(o-arsonophenyl)azo]-3-hydroxy-2,7-naphthalenedisulfonate, C16H11AsN2Na2O10 S2.
Thorium nitrate (0.005 mol/L) VS
Thorium nitrate R dissolved in water to contain 2.401 g of Th(NO3)4 in 1000 mL.
Method of standardization. Ascertain the exact concentration of the 0.005 mol/L solution in the following manner: transfer 0.050 g, accurately weighed, of sodium fluoride R, previously dried, to a flask and dissolve in sufficient water to produce 250 mL. To 20.0 mL of this solution add 0.6 mL of sodium alizarinsulfonate (1 g/L) TS and then, by drops, sodium hydroxide (0.1 mol/L) VS until the colour changes from pink to yellow. Add 5 mL of acetate buffer, pH 3.0, TS and titrate with the thorium nitrate solution until the yellow colour changes to pinkish yellow. Each 0.8398 mg of sodium fluoride is equivalent to 1 mL of thorium nitrate (0.005 mol/L) VS.
Thorium nitrate R
Th(NO3)4,4H2O.
Description. White, slightly deliquescent crystals.
Solubility. Very soluble in water and ethanol (~750 g/L) TS.
Thymine R
5-methylpyrimidine-2,4(1H,3H)-dione; C5H6N2O2.
A commercially available reagent of suitable grade.
Description. Short needles or plates.
Solubility. Slightly soluble in cold water, soluble in hot water. It dissolves in dilute solution of alkali hydroxyde.
Thymol blue R
Thymolsulfonphthalein, C27H30O5S (SRIP, 1963, p. 207).
Thymol blue/dimethylformamide TS
Procedure. Dissolve 0.3 g of thymol blue R in sufficient dimethylformamide R to produce 100 mL.
Thymol blue/ethanol TS
Procedure. Dissolve 0.1 g of thymol blue R in sufficient ethanol (~750 g/L) TS to produce 100 mL; filter if necessary.
Thymol blue/methanol TS
Procedure. Dissolve 0.3 g of thymol blue R in sufficient methanol R to produce 100 mL.
Thymol R
C10H14O.
Description. Colourless, often large crystals, or a white, crystalline powder; odour, aromatic, resembling that of thyme.
Solubility. Soluble in about 1000 parts of water, in 1 part of ethanol (~750 g/L) TS and in 1.5 parts of ether R.
Melting range. Between 48 and 51 °C; when the melted substance is cooled it remains liquid at a considerably lower temperature.
Residue on volatilization. Volatilize 2 g on a water-bath and dry to constant weight at 105 °C; it leaves a residue of not more than 0.5 mg/g.
Storage. Store in tightly closed containers, protected from light.
Thymol TS1
Procedure. Dissolve 0.225 g of thymol R in sufficient carbon tetrachloride R to produce 100 mL.
Thymol TS2
Procedure. Dilute 10 mL of thymol TS1 to 100 mL with carbon tetrachloride R.
Thymol TS3
Procedure. Dilute 10 mL of thymol TS1 to 150 mL with carbon tetrachloride R.
Thymolphthalein R
C28H30O4 (SRIP, 1963, p. 207).
Thymolphthalein/dimethylformamide TS
Procedure. Dissolve 0.1 g of thymolphthalein R in sufficient dimethylformamide R to produce 100 mL.
Thymolphthalein/ethanol TS
Procedure. Dissolve 0.1 g of thymolphthalein R in 100 mL of ethanol (~750 g/L) TS and filter if necessary.
Titan yellow paper R
Impregnate filter paper with titan yellow TS. Allow to dry at room temperature.
Titan yellow R
C28H19N5Na2O6S4 (SRIP, 1963, p. 208).
Titan yellow TS
Procedure. Dissolve 0.05 g of titan yellow R in sufficient water to produce 100 mL.
Titanium dioxide R
TiO2.
Description. A white powder; odourless.
Solubility. Practically insoluble in water; slowly soluble, when heated, in sulfuric acid (~1760 g/L) TS.
Titanium dioxide/sulfuric acid TS
Procedure. To 0.1 g of titanium dioxide R add 100 mL of sulfuric acid (~1760 g/L) TS. Heat cautiously with occasional stirring until a clear solution is effected and fumes are evolved; cool.
Storage. Store in glass-stoppered bottles.
Titanium trichloride (0.1 mol/L) VS
Procedure. Dilute 100 mL of titanium trichloride R with 200 mL of hydrochloric acid (~250 g/L) TS and add sufficient carbon-dioxide-free water R to produce 1000 mL.
Method of standardization. Ascertain the exact concentration immediately before use. With the solution titrate 25 mL of ferric ammonium sulfate (0.1 mol/L) VS acidified with sulfuric acid (~100 g/L) TS in an atmosphere of carbon dioxide R, adding ammonium thiocyanate (75 g/L) TS just before the end-point as indicator. Each mL of ferric ammonium sulfate (0.1 mol/L) VS is equivalent to 15.43 mg of TiCl3.
Titanium trichloride R
A solution of titanium trichloride containing about 15% of TiCl3 (SRIP, 1963, p. 208).
Mass density. ρ20 = ~1.2 kg/L.
Toluene R
C7H8 (SRIP, 1963, p. 209).
4-Toluenesulfonamide R
C7H9NO2S.
Melting range. 135–137 °C.
4-Toluenesulfonic acid R
C7H8O3S,H2O. Contains not less than 98.0% of C7H8O3S.
Description. Colourless crystals or a white, crystalline powder.
Solubility. Soluble in water, ethanol (~750 g/L) TS and ether R.
Melting range. 100–105 °C.
Sulfated ash. Not more than 1.0 mg/g.
Assay. Dissolve 0.8 g, accurately weighed, in 50 mL of water and titrate with sodium hydroxide (0.1 mol/L) VS using phenolphthalein/ethanol TS as indicator. Each mL of sodium hydroxide (0.1 mol/L) VS is equivalent to 19.02 mg of C7H8O3S,H2O.
4-Toluenesulfonic acid/ethanol TS
Procedure. Dissolve 20 g of 4-toluenesulfonic acid R in sufficient ethanol (~750 g/L) TS to produce 100 mL.
Tosylchloramide sodium (15 g/L) TS
A solution of tosylchloramide sodium R containing about 16 g of C7H7ClNNaO2S per litre.
Tosylchloramide sodium R
C7H7ClNNaO2S,3H2O. Contains not less than 98.0% of C7H7ClNNaO2S,3H2 O.
Description. White crystals or a white, crystalline powder; odour, resembling that of chlorine.
Solubility. Soluble in 7 parts of water and in 2 parts of boiling water; soluble in ethanol (~750 g/L) TS; insoluble in ether R.
Sodium chloride. Treat 1.0 g with 15 mL of dehydrated ethanol R without the aid of heat and filter; it leaves a residue of not more than 15 mg.
Assay. Dissolve 0.4 g, accurately weighed, in 50 mL of water, placed in a glass-stoppered vessel. Add 10 mL of potassium iodide (80 g/L) TS and 5 mL of sulfuric acid (~100 g/L) TS. Allow to stand for 10 minutes and titrate the liberated iodine with sodium thiosulfate (0.1 mol/L) VS. Each mL of sodium thiosulfate (0.1 mol/L) VS is equivalent to 14.08 mg of C7H7ClNNaO2S,3H2O.
Note: Tosylychloramide sodium R is efflorescent.
Tributyl phosphate R
C12H27O4P.
Description. A clear, colourless liquid.
Miscibility. Slightly miscible with water; miscible with most organic solvents.
Mass density. ρ20 = about 0.98 kg/L.
Note: Before use, wash the reagent three times as follows: shake 60 mL with 10 mL of a solution containing 1 g of sodium chloride R and 0.1 g of disodium hydrogen phosphate R.
Trichloroacetic acid R
C2HCl3O2. (SRIP, 1963, p. 209).
Trichloroethylene R
C2HCl3.
Description. A colourless or pale blue, clear, mobile liquid; odour, characteristic, resembling that of chloroform.
Miscibility. Almost immiscible with water; miscible with dehydrated ethanol R and ether R.
Trichlorotrifluoroethane R
1,1,2-Trichloro-1,2,2-trifluoroethane. C2Cl3F3.
Description. A colourless, volatile liquid.
Miscibility. Immiscible with water; miscible with acetone R and ether R.
Trichlorotrifluoroethane TS
Procedure. Mix 0.05 μL of trichlorotrifluoroethane R with 1.0 mL of dichloromethane R (other suitable solvents can be used).
Triethylamine R
C6H15N.
Description. A colourless liquid; odour, ammoniacal.
Boiling range. 89–90 °C.
Mass density. ρ20 = about 0.73 kg/L.
Refractive index. 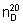 = 1.4003.
= 1.4003.
Triethylenediamine R
1,4-Diazabicyclo[2.2.2]octane; C6H12N2.
Description. Hygroscopic crystals.
Melting temperature. About 158 °C.
Storage. Store in a tightly closed container.
Trifluoroacetic acid R. C2HF3O2
A commercially available reagent of suitable grade.
Triketohydrindene hydrate (1 g/L) TS
A solution of triketohydrindene hydrate R containing about 1 g of C9H4O3 per litre.
Triketohydrindene hydrate R
Ninhydrin, C9H4O3,H2O (SRIP, 1963, p. 210).
Triketohydrindene/butanol TS
Procedure. Dissolve 0.1 g of triketohydrindene hydrate R in sufficient 1-butanol R previously saturated with water to produce 100 mL.
Triketohydrindene/butanol/acetic acid TS
Procedure. Prepare a 20 mg/mL solution of triketohydrindene hydrate R in a mixture of 95 volumes of 1-butanol R and 5 volumes of acetic acid (~120 g/L) TS.
Triketohydrindene/cadmium TS
Procedure. Dissolve 0.050 g of cadmium acetate R in a mixture of 5 mL of water and 1 mL of glacial acetic acid R and add sufficient ethylmethylketone R to produce 50 mL. Dissolve 20 mg of triketohydrindene hydrate R in 10 mL of this solution.
Note: Prepare immediately before use.
Triketohydrindene/ethanol TS
Procedure. Prepare a saturated solution of triketohydrindene hydrate R in ethanol (~750 g/L) TS.
Triketohydrindene/methanol TS
Procedure. Dissolve 1.0 g of triketohydrindene hydrate R in sufficient methanol R to produce 100 mL.
Note. Triketohydrindene/methanol TS must be freshly prepared.
Triketohydrindene/pyridine/acetone TS
Procedure. Dissolve 0.25 g of triketohydrindene hydrate R in 100 mL of a mixture of equal volumes of pyridine R and acetone R.
Triketohydrindene/pyridine/butanol TS
Procedure. Dissolve 1 g of triketohydrindene hydrate R in 1 mL of pyridine R and dilute with sufficient 1-butanol R to produce 100 mL.
Note: It should be freshly prepared.
Triketohydrindene/sodium metabisulfite TS
Procedure. Dissolve 3 g of triketohydrindene hydrate R in 100 mL of a solution containing 4.55 g of sodium metabisulfite R in 100 mL of water.
Triketohydrindene/stannous chloride TS
Procedure. Dissolve 4 g of triketohydrindene hydrate R in 100 mL of ethylene glycol monomethyl ether R. Shake gently with 1 g of cation exchange resin (300 μm–840 μm) and filter (solution A). Dissolve 0.16 g of stannous chloride R in 100 mL of acetate buffer, pH 5.5, TS (solution B). Immediately before use mix equal volumes of the two solutions.
2,4,4 Trimethylpentan-2-amine R
C8H19N
Molecular weight. 129.3.
Other name. 2-Amino-2,4,4-trimethylpentane; 1,1,3,3-Tetramethylbutylamine
Description. Clear, colourless liquid.
Relative density![]() . About 0.805.
. About 0.805.
Boiling point. About 140 °C.
2,2,4-Trimethylpentane R
iso -Octane; C8H18 (SRIP, 1963, p. 129).
Trimethylpyridine (50 g/L) TS
A solution of trimethylpyridine R containing about 50 g of C8H11N per litre.
Trimethylpyridine R
C8H11N. 2,4,6-Trimethylpyridine; sym. collidine.
Description. Liquid; odour, aromatic.
Miscibility. More miscible with cold water than with hot water; miscible with ethanol (~750 g/L) TS; ether R and methanol R.
Relative density. 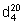 = 0.914.
= 0.914.
Refractive index. 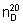 = 1.498.
= 1.498.
Trinitrophenol (7 g/L) TS
A solution of trinitrophenol R containing 7 g of C6H3N3O7 per litre.
Trinitrophenol R
C6H3N3O7 (SRIP, 1963, p. 211).
Trinitrophenol, alkaline, TS
Procedure. Mix 20 mL of a 10 mg/mL solution of trinitrophenol R with 10 mL of a 50 mg/mL solution of sodium hydroxide R, dilute with water to 100 mL and mix.
Note: After preparation alkaline trinitrophenol TS should be used for only 48 hours.
Trinitrophenol/ethanol TS
Procedure. Dissolve 33 g of trinitrophenol R in sufficient ethanol (~750 g/L) TS to produce 1000 mL.
Triphenylantimony R
C18H15Sb.
Melting temperature. About 55 °C.
Triphenylmethanol R
Triphenylcarbinol; C19H16O.
A commercialy available reagent of suitable grade.
Description. Colourless crystals.
Solubility. Practically insoluble in water, freely soluble in ethanol (~750 g/L) TS (ethanol (95%) R).
Trisodium orthophosphate (2 g/L) TS
A solution of trisodium orthophosphate R containing about 2 g of Na3PO4 per litre.
Trisodium orthophosphate R
Na3PO4,12H2O.
Description. Colourless crystals or a white, crystalline powder.
Solubility. Freely soluble in water; practically insoluble in ethanol (~750 g/L) TS and carbon disulfide R.
Tyrosine R
C9H11NO3 (SRIP, 1963, p. 212).