Monographs: Pharmaceutical substances: Loperamide hydrochloride (Loperamidi hydrochloridum)
Molecular formula. C29H33ClN2O2,HCl
Relative molecular mass. 513.5
Graphic formula.
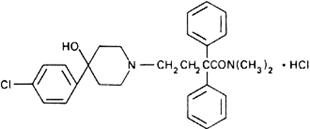
Chemical name. 4-(p-Chlorophenyl)-4-hydroxy-N,N-dimethyl-α,α-diphenyl-1-piperidinebutyramide monohydrochloride; 4-(4-chlorophenyl)-4-hydroxy-N,N-dimethyl-α,α-diphenyl-1-piperidinebutanamide monohydrochloride; CAS Reg. No. 34552-83-5.
Description. A white to slightly yellowish powder.
Solubility. Slightly soluble in water and in dilute acids; freely soluble in methanol R.
Category. Antidiarrhoeal drug.
Storage. Loperamide hydrochloride should be kept in a well-closed container.
Requirements
Definition. Loperamide hydrochloride contains not less than 98.0% and not more than 102.0% of C29H33ClN2O2,HCl, calculated with reference to the dried substance.
Identity tests
• Either tests A and D or tests B, C and D may be applied.
A. Carry out the examination as described under 1.7 Spectrophotometry in the infrared region. The infrared absorption spectrum is concordant with the spectrum obtained from loperamide hydrochloride RS or with the reference spectrum of loperamide hydrochloride.
B. Transfer about 0.04 g, accurately weighed, to a 100-mL volumetric flask, dissolve in about 50 mL of 2-propanol R, add 10 mL of hydrochloric acid (0.1 mol/l) VS and dilute to volume with 2-propanol R. The absorption spectrum of this solution against a solvent cell containing the same solvent mixture, when observed between 230 nm and 350 nm, is qualitatively similar to that of a solution of loperamide hydrochloride RS concurrently examined (maxima occur at about 253 nm, 259 nm, 265 nm, and 273 nm). The absorbances of the solutions at the respective maxima do not differ from each other by more than 3%.
C. Melting temperature, about 224 °C with decomposition.
D. A 10 mg/mL solution yields reaction A described under 2.1 General identification tests as characteristic of chlorides.
Sulfated ash. Not more than 2.0 mg/g.
Loss on drying. Dry at 80°C under reduced pressure (not exceeding 0.6 kPa or about 5 mm of mercury) for 4 hours; it loses not more than 5.0 mg/g.
Related substances. Carry out the test as described under 1.14.1 Chromatography, Thin-layer chromatography, using silica gel R1 as the coating substance and a mixture of 85 volumes of chloroform R, 10 volumes of methanol R, and 5 volumes of formic acid (~1080 g/l) TS as the mobile phase. Apply separately to the plate 10 μl of each of 2 solutions in chloroform R containing (A) 10 mg of the test substance per mL and (B) 0.10 mg of the test substance per mL. After removing the plate from the chromatographic chamber, allow it to dry in air and expose it to iodine vapours. Examine the chromatogram in daylight. Any spot obtained with solution A, other than the principal spot, is not more intense than that obtained with solution B.
Assay. Dissolve 0.400 g in 50 mL of dehydrated ethanol R and add 5.0 mL of hydrochloric acid (0.01 mol/L) VS. Carry out a potentiometric titration, using sodium hydroxide (0.1 mol/L) VS, as described under 2.6 Non-aqueous titration. Read the volume added between the two points of inflexion.
1 mL of sodium hydroxide (0.1 mol/L) VS is equivalent to 51.35 mg of C29H33ClN2O2,HCl.