Monographs: Pharmaceutical substances: Paromomycin sulfate (Paromomycini sulfas)
Molecular formula. C23H45N5O14,xH2SO4
Graphic formula.
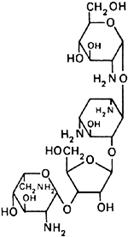
Chemical name. O-2,6-Diamino-2,6-dideoxy-β-L-idopyranosyl-(1→3)-O-β-D-ribofuranosyl-(1→5)-O-[2-amino-2-deoxy-α-D-glucopyranosyl-(1→4)]-2-deoxystreptamine sulfate (salt); O-2-amino-2-deoxy-α-D-glucopyranosyl-(1→4)-O-[O-2,6-diamino-2,6-dideoxy-β-L-idopyranosyl-(1→3)-β-D-ribofuranosyl-(1→5)]-2-deoxy-D-streptamine sulfate (salt); CAS Reg. No. 1263-89-4.
Description. A creamy white to light yellow powder; odourless or almost odourless.
Solubility. Very soluble in water; practically insoluble in ethanol (~750 g/l) TS and ether R.
Category. Antiamoebic drug.
Storage. Paromomycin sulfate should be kept in a tightly closed container, protected from light.
Additional information. Paromomycin sulfate is very hygroscopic. Even in the absence of light, it is gradually degraded on exposure to a humid atmosphere, the decomposition being faster at higher temperatures.
Requirements
Definition. Paromomycin sulfate contains not less than 675 International Units of paromomycin per mg, calculated with reference to the dried substance.
Identity tests
A. Carry out the test as described under 1.14.1 Chromatography, Thin-layer chromatography, using silica gel R3 as the coating substance and a freshly prepared ammonium acetate (40 g/l) TS as the mobile phase. Apply separately to the plate 1 μl of each of 2 solutions containing (A) 20 mg of the test substance per mL and (B) 20 mg of paromomycin sulfate RS per mL. After removing the plate from the chromatographic chamber, allow it to dry in air for 10 minutes, heat it at 105°C for 1 hour, and spray it with triketohydrindene/butanol TS. Heat it again at 105°C for 5 minutes and examine the chromatogram in daylight. The principal red spot obtained with solution A corresponds in position and appearance with that obtained with solution B.
B. A 0.05 g/mL solution yields reaction A described under 2.1 General identification tests as characteristic of sulfates.
Specific optical rotation. Use a 50 mg/mL solution and calculate with reference to the dried substance; 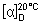 = +50° to +55°.
= +50° to +55°.
Sulfated ash. Not more than 20 mg/g.
Loss on drying. Dry to constant weight at 50°C under reduced pressure (not exceeding 0.6 kPa or about 5 mm of mercury); it loses not more than 50 mg/g.
pH value. pH of a 30 mg/mL solution in carbon-dioxide-free water R, 5.0-7.5.
Assay. Carry out the assay as described under 3.1 Microbiological assay of antibiotics, using either (a) Bacillus subtilis (NCTC 10400) as the test organism, culture medium Cm1 with a final pH of 8.0, sterile phosphate buffer pH 7.8, TS, an appropriate concentration of paromomycin (usually between 1 and 4 IU per mL), and an incubation temperature of 37-39°C; or (b) Bacillus subtilis (ATCC 6633) as the test organism, culture medium Cm1 with a final pH of 7.8, sterile phosphate buffer pH 8.0, TS1 or TS2, an appropriate concentration of paromomycin (usually between 2 and 8 IU per mL), and an incubation temperature of 36-38°C. The precision of the assay is such that the fiducial limits of error of the estimated potency (P = 0.95) are not less than 95% and not more than 105% of the estimated potency. The upper fiducial limit of error of the estimated potency (P = 0.95) is not less than 675 IU of paromomycin per mg, calculated with reference to the dried substance.