Monographs: Pharmaceutical substances: Prednisolone sodium phosphate (Prednisoloni natrii phosphas)
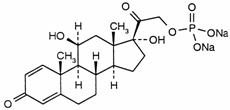
C21H27Na2O8P
Relative molecular mass. 484.4
Chemical name. 11β,17,21-Trihydroxypregna-1,4-diene-3,20-dione 21-(disodium phosphate); (11β) 11,17-dihydroxy-21-(phosphonooxy)-pregna-1,4-diene-3,20-dione disodium salt; CAS Reg. No. 125-02-0.
Description. A white to light yellow, crystalline powder or granules.
Solubility. Freely soluble in water; soluble in methanol R; very slightly soluble in ethanol (~750 g/l) TS and acetone R.
Category. Corticosteroid.
Storage. Prednisolone sodium phosphate should be kept in a tightly closed container, protected from light.
Labelling. The designation Prednisolone sodium phosphate for sterile noninjectable use indicates that the substance complies with the additional requirement and may be used for sterile applications. Expiry date.
Additional information. Prednisolone sodium phosphate is hygroscopic.
Requirements
Prednisolone sodium phosphate contains not less than 96.0% and not more than 103.0% of C21H27Na2O8P, calculated with reference to the anhydrous substance.
Identity tests
• Either tests A, D, and E or tests B, C, D, and E may be applied.
A. Carry out the examination as described under 1.7 Spectrophotometry in the infrared region. The infrared absorption spectrum is concordant with the spectrum obtained from prednisolone sodium phosphate RS or with the reference spectrum of prednisolone sodium phosphate.
B. Carry out the test as described under 1.14.1 Chromatography, Thin-layer chromatography, using silica gel R1 as the coating substance and a freshly prepared mixture of 3 volumes of 1-butanol R, 1 volume of acetic anhydride R, and 1 volume of water as the mobile phase. Apply separately to the plate 2μl of each of 4 solutions in methanol R containing (A) 2.5mg of Prednisolone sodium phosphate per mL, (B) 2.5mg of prednisolone sodium phosphate RS per mL, (C) a mixture of equal volumes of solutions A and B, and (D) equal volumes of solution A and a solution of 2.5 mg of dexamethasone sodium phosphate RS per mL of methanol R. After removing the plate from the chromatographic chamber, allow it to dry in air until the solvents have evaporated, spray with a mixture of 10 mL of sulfuric acid (~1760 g/l) TS and 90 mL of ethanol (~750 g/l) TS, heat at 120 °C for 10 minutes, allow to cool, and examine the chromatogram in ultraviolet light (365 nm).
The principal spot obtained with solution A corresponds in position, appearance, and intensity with that obtained with solution B. The principal spot obtained with solution C appears as a single compact spot, whereas the chromatogram obtained with solution D shows two spots which may not be completely separated.
C. To about 2mg add 2 mL of sulfuric acid (~1760 g/l) TS and shake to dissolve; an intense red colour is produced within 5 minutes. Examine under ultraviolet light (365 nm); a reddish brown fluorescence is observed. Add the solution to 10 mL of water and mix; the colour fades and a greenish yellow fluorescence is produced.
D. When tested for sodium as described under 2.1 General identification tests, it yields the characteristic reactions. If reaction B is to be used, prepare a 20 mg/mL solution.
E. To 1 mL of a 20 mg/mL solution add 3 mL of nitric acid (~130 g/l) TS; it yields reaction A described under 2.1 General identification tests as characteristic of orthophosphates.
Specific optical rotation. Use a 10 mg/mL solution and calculate with reference to the anhydrous substance; 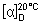 = +95° to +102°.
= +95° to +102°.
Clarity and colour of solution. A solution of 0.5 g in 10 mL of carbon-dioxide-free water R is clear and not more intensely coloured than standard colour Bn1 when compared as described under 1.11.1 Colour of liquids.
Water. Determine as described under 2.8 Determination of water by the Karl Fischer method, Method A, using about 0.2 g of the substance; not more than 0.080 g/g.
pH value. pH of a 0.05 g/mL solution in carbon-dioxide-free water R, 7.5-9.0.
Inorganic phosphates. Dissolve 0.050 g in sufficient water to produce 100 mL. To 10ml add 5ml of ammonium molybdate/vanadate TS, mix and allow to stand for 5 minutes; any yellow colour produced is not more intense than that of a reference solution prepared similarly using 10 mL of phosphate standard (5μg/mL) TS.
Free prednisolone and other related substances
• Either test A or test B may be applied.
A. Carry out the test as described under 1.14.1 Chromatography, High-performance liquid chromatography, using a stainless steel column (15cm × 4.6mm) packed with particles of silica gel, the surface of which has been modified with chemically bonded octadecylsilyl groups (5μm). As the mobile phase, use a mixture prepared as follows: weigh 1.36 g of potassium dihydrogen phosphate R and 0.60 g of hexylamine R, transfer to a 250-mL conical flask, mix, and allow to stand for 10 minutes, and then dissolve in 185 mL of water. Add 65 mL of acetonitrile R, mix, and filter.
Prepare the following solutions in the mobile phase: solution (A) 2.5mg of Prednisolone sodium phosphate per mL; solution (B) 2.5 mg of prednisolone sodium phosphate RS and 2.5 mg of prednisolone RS per mL, dilute 1.0ml of this solution to 25 mL with the mobile phase; and for solution (C) dilute 1.0 mL of solution A to 50 mL with the mobile phase.
Operate with a flow rate of 1.0 mL per minute. As a detector use an ultraviolet spectrophotometer set at a wavelength of 254nm.
Inject 20μl of solution B. Adjust the sensitivity of the system so that the heights of the principal peaks in the chromatogram obtained with solution B are 70-90% of the full scale of the recorder. The retention times for prednisolone sodium phosphate are about 6.5 minutes, and for prednisolone about 8.5 minutes. The test is not valid unless the resolution between the peaks corresponding to prednisolone sodium phosphate and prednisolone is not less than 4.5.
Inject alternately 20μl each of solutions A and C. Continue the chromatography for 3 times the retention time of the principal peak.
Measure the areas of the peak responses obtained in the chromatograms from solutions A and C, and calculate the contents of free prednisolone and other related substances as a percentage. In the chromatogram obtained from solution A, the area of any peak, other than the principal peak, is not greater than that of the principal peak obtained with solution C (2.0%), and not more than one such peak has an area greater than half the area of the principal peak obtained with solution C (1.0%). The sum of the areas of all the peaks, other than the principal peak, is not greater than 1.5 times the area of the principal peak obtained with solution C (3.0%). Disregard any peak due to the solvent and any peak with an area less than 0.025 times the area of the principal peak obtained with solution C.
B. Carry out the test as described under 1.14.1 Chromatography, Thin-layer chromatography, using silica gel R1 as the coating substance and methanol R as the mobile phase. Apply separately to the plate 2μl of each of 2 solutions in methanol R containing (A) 10 mg of Prednisolone sodium phosphate per mL, and (B) 0.20 mg of prednisolone RS per mL. After removing the plate from the chromatographic chamber, allow it to dry in air for 5 minutes, spray with a solution of 3 g of zinc chloride R in 10 mL of methanol R, heat at about 125 °C for 1 hour, and examine the chromatogram in ultraviolet light (365 nm).
Any spot obtained with solution A, other than the principal spot, is not more intense than that obtained with solution B (2.0%).
Assay. Dissolve about 0.1 g, accurately weighed, in sufficient water to produce 100ml. Dilute 5 mL to 250 mL with water and measure the absorbance of this solution in a 1-cm layer at the maximum at about 247nm.
Calculate the percentage content of C21H27Na2O8P using the absorptivity value of 31.2 (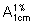 = 312).
= 312).
Additional requirement for sterile non-injectable Prednisolone sodium phosphate
Complies with 3.2 Test for sterility.