Monographs: Pharmaceutical substances: Procaine hydrochloride (Procaini hydrochloridum)
Molecular formula. C13H20N2O2,HCl
Relative molecular mass. 272.8
Graphic formula.
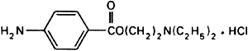
Chemical name. 2-(Diethylamino)ethyl p-aminobenzoate monohydrochloride; 2-(diethylamino)ethyl 4-aminobenzoate monohydrochloride; CAS Reg. No. 51-05-8.
Description. Colourless crystals or a white, crystalline powder; odourless.
Solubility. Soluble in 1 part of water and in 25 parts of ethanol (~750 g/l) TS; practically insoluble in ether R.
Category. Local anaesthetic.
Storage. Procaine hydrochloride should be kept in a tightly closed container, protected from light.
Additional information. Procaine hydrochloride causes local numbness after being placed on the tongue. Even in the absence of light, Procaine hydrochloride is gradually degraded on exposure to a humid atmosphere, the decomposition being faster at higher temperatures.
Requirements
Definition. Procaine hydrochloride contains not less than 99.0% and not more than 101.0% of C13H20N2O2,HCl, calculated with reference to the dried substance.
Identity tests
• Either tests A and D or tests B, C, and D may be applied.
A. Carry out the examination as described under 1.7 Spectrophotometry in the infrared region. The infrared absorption spectrum is concordant with the spectrum obtained from procaine hydrochloride RS or with the reference spectrum of procaine hydrochloride.
B. About 0.05 g yields the reaction described for the identification of primary aromatic amines under 2.1 General identification tests, producing a vivid red precipitate.
C. Dissolve 0.05 g in 5 mL of water, add 5 drops of sulfuric acid (~100 g/l) TS and 2 drops of potassium permanganate (0.02 mol/l) VS; the violet colour produced disappears quickly.
D. A 0.05 g/mL solution yields reaction B described under 2.1 General identification tests as characteristic of chlorides.
Melting range. 154-158°C.
Heavy metals. Use 1.0 g for the preparation of the test solution as described under 2.2.3 Limit test for heavy metals. Procedure 1; determine the heavy metals content according to Method A; not more than 20 μg/g.
Clarity and colour of solution. A solution of 1.0 g in 10 mL of carbon-dioxide-free water R is clear and colourless.
Sulfated ash. Not more than 1.5 mg/g.
Loss on drying. Dry to constant weight at 105°C; it loses not more than 10 mg/g.
pH value. pH of a 10 mg/mL solution in carbon-dioxide-free water R, 5.0-6.5.
Related substances. Carry out the test as described under 1.14.1 Chromatography, Thin-layer chromatography, using silica gel R2 as the coating substance and a mixture of 80 volumes of dibutyl ether R, 16 volumes of hexane R, and 4 volumes of glacial acetic acid R as the mobile phase. Apply separately to the plate 5 μl of each of 2 solutions containing (A) 0.10 g of the test substance per mL and (B) 0.050 mg of 4-aminobenzoic acid R per mL. After removing the plate from the chromatographic chamber, allow it to dry at 105°C for 10 minutes, and examine the chromatogram in ultraviolet light (254 nm). Any spot obtained with solution A, other than the principal spot, is not more intense than that obtained with solution B. The principal spot remains at the point of application.
Assay. Carry out the assay as described under 2.7 Nitrite titration; dissolve about 0.5 g, accurately weighed, in 50 mL of hydrochloric acid (~70 g/l) TS, add 0.1 g of potassium bromide R, and titrate with sodium nitrite (0.1 mol/l) VS. Each mL of sodium nitrite (0.1 mol/l) VS is equivalent to 27.28 mg of C13H20N2O2,HCl.