Monographs: Pharmaceutical substances: Cyclophosphamide (Cyclophosphamidum)
Molecular formula. C7H15Cl2N2O2P,H2O
Relative molecular mass. 279.1
Graphic formula.
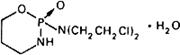
Chemical name. 2-[Bis(2-chloroethyl)amino]tetrahydro-2H-1,3,2-oxazaphosphorine 2-oxide monohydrate; N,N-bis(2-chloroethyl)tetrahydro-2H-1,3,2-oxazaphosphorin-2-amine 2-oxide monohydrate; CAS Reg. No. 6055-19-2 (monohydrate).
Other name. Cyclophosphanum.
Description. A white, crystalline powder.
Solubility. Soluble in water; freely soluble in ethanol (~750 g/l) TS; slightly soluble in ether R.
Category. Cytotoxic drug.
Storage. Cyclophosphamide should be kept in a tightly closed container and stored at a temperature between 2 and 30 °C.
Additional information. CAUTION: Cyclophosphamide must be handled with care, avoiding contact with the skin and inhalation of airborne particles.
Requirements
Definition. Cyclophosphamide contains not less than 98.0% and not more than 101.0% of C7H15Cl2N2O2P, calculated with reference to the anhydrous substance.
Identity tests
A. Dissolve 0.1 g in 10 mL of water and add 5 mL of silver nitrate (40 g/l) TS; no precipitate is produced. Boil; a white precipitate is produced, which is insoluble in nitric acid (~130 g/l) TS but soluble in ammonia (~100 g/l) TS from which it is reprecipitated by the addition of nitric acid (~130 g/l) TS.
B. Dissolve 20 mg in 1 mL of sulfuric acid (~100 g/l) TS and heat until white fumes are evolved. After cooling, add 5 mL of water and shake. Neutralize with ammonia (~100 g/l) TS, then acidify with nitric acid (~130 g/l) TS; this test yields reaction A described under 2.1 General identification tests as characteristic of orthophosphates.
Melting range. 49-53 °C, determined without previous drying.
Clarity and colour of solution. A solution of 0.20 g in 10 mL of water is clear and colourless.
Water. Determine as described under 2.8 Determination of water by the Karl Fischer method, Method A, using about 0.3 g of the substance; the water content is not less than 55 mg/g and not more than 70 mg/g.
pH value. pH of a 20 mg/mL solution in carbon-dioxide-free water R, determined 30 minutes after its preparation, 4.0-7.0.
Related substances. Carry out the test as described under 1.14.1 Chromatography, Thin-layer chromatography, using silica gel R1 as the coating substance and a mixture of 50 volumes of benzene R, 25 volumes of chloroform R, and 25 volumes of methanol R as the mobile phase. Apply separately to the plate 10 μl of each of 2 solutions: (A) 25 mg of the test substance per mL of chloroform R and (B) 0.125 g of the test substance dissolved in 5.0 mL of water, boiled under a reflux condenser for 30 minutes, and then cooled to 20 °C. After removing the plate from the chromatographic chamber, allow it to dry in air and spray it with triketohydrindene/methanol TS. Examine the chromatogram in daylight. With solution B, a pale violet spot is obtained at an Rf value between 0.10 and 0.25; other spots could also appear. With solution A, a brown-violet spot is obtained with an Rr value between 3.50 and 5.50, and no other spot is obtained above this spot.
Assay. To about 0.2 g, accurately weighed, add 20 mL of potassium hydroxide/ethanol (0.5 mol/l) VS. Boil under a reflux condenser for 1 hour, cool, then add 30 mL of water, 3 mL of nitric acid (~1000 g/l) TS and 20.0 mL of silver nitrate (0.1 mol/l) VS. Shake the flask, add 5 mL of diethyl phthalate R and titrate the excess of silver nitrate with ammonium thiocyanate (0.1 mol/l) VS, using 5 mL of ferric ammonium sulfate (45 g/l) TS as indicator. Repeat the operation without the substance being examined and make any necessary corrections. Each mL of silver nitrate (0.1 mol/l) VS is equivalent to 13.05 mg of C7H15Cl2N2O2P.