Monographs: Pharmaceutical substances: Cycloserine (Cycloserinum)
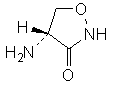
C3H6N2O2
Relative molecular mass. 102.1.
Chemical name. (4R)-4-aminoisoxazolidin-3-one; (4R)-4-amino-1,2-oxazolidin-3-one; (+)-4-amino-3-isoxazolidinone; CAS Reg. No. 68-41-7.
Description. A white or pale yellow, crystalline, powder.
Solubility. Freely soluble in water; slightly soluble in methanol R and propylene glycol R; very slightly soluble in ethanol (~750 g/L) TS; practically insoluble in dichloromethane R.
Category. Antibacterial; tuberculostatic.
Storage. Cycloserine should be kept in a tightly closed container.
Additional information. Cycloserine is slightly hygroscopic and degrades upon exposure to a humid atmosphere, decomposition being faster at higher temperatures.
Requirements
Definition. Cycloserine is an analogue of the amino acid D-alanine with broad-spectrum antibiotic and glycinergic activities produced by Streptomyces garyphalus and Streptomyces orchidaceus or obtained by synthesis.
Cycloserine contains not less than 98.5% and not more than 101.5% of cycloserine (C3H6N2O2), calculated with reference to the dried substance.
Identity tests
- Either tests A and C, or tests B and C or test D alone may be applied.
A. Carry out test A.1 or, where UV detection is not available, test A.2.
A.1 Carry out the test as described under 1.14.1 Chromatography, Thin-layer chromatography using silica gel R6 as the coating substance and a mixture of 4 volumes of 1-butanol R, 1 volume of glacial acetic acid R and 2 volumes of water R as the mobile phase. Apply separately to the plate 10 µL of each of the following two solutions. For solution (A) dissolve 20 mg of the test substance in 0.5 mL of water R, add 4.5 mL of methanol R and shake. For solution (B) use 20 mg of cycloserine RS prepared in the same manner. After removing the plate from the chromatographic chamber allow it to dry exhaustively in a current of air. Examine the chromatogram in ultraviolet light (254 nm).
The principal spot obtained with solution (A) corresponds in position, appearance and intensity with that obtained with solution (B).
A.2 Carry out the test as described under 1.14.1 Chromatography, Thin-layer chromatography using the conditions described above under test A.1 but using silica gel R5 as the coating substance. After removing the plate from the chromatographic chamber allow it to dry in a current of air and place the plate in a chamber with iodine vapours. Examine the chromatogram in daylight.
The principal spot obtained with solution (A) corresponds in position, appearance and intensity with that obtained with solution (B).
B. Dissolve about 1 mg in 10 mL of sodium hydroxide (~4 g/L) TS. To 1 mL of the resulting solution add 3 mL of acetic acid (~60 g/L) TS and 1 mL of a recently prepared mixture of equal volumes of a 40 mg/mL solution of sodium nitroprusside R and sodium hydroxide (~200 g/L) TS; a blue colour gradually develops.
C. The absorption spectrum (1.6) of a freshly prepared 25 μg/mL solution in hydrochloric acid (~3.647 g/L) TS, when observed between 215 nm and 360 nm, exhibits a maximum at about 219 nm; the specific absorbance (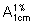 ) is between 327 and 361.
) is between 327 and 361.
D. Carry out the examination as described under 1.7 Spectrophotometry in the infrared region. The infrared absorption spectrum is concordant with the spectrum obtained from cycloserine RS or with the reference spectrum of cycloserine.
Specific optical rotation (1.4). Use a 50 mg/mL solution in sodium hydroxide (~80 g/L) TS and calculate with reference to the dried substance; 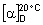 = +108° to +114°.
= +108° to +114°.
Heavy metals. Use 2.0 g for the preparation of the test solution as described under 2.2.3 Limit test for heavy metals, procedure 3; determine the heavy metals content according to method A; not more than 10 µg/g.
Sulfated ash (2.3). Not more than 5.0 mg/g.
Loss on drying. Dry at 60 °C under reduced pressure (not exceeding 0.6 kPa or about 5 mm of mercury) for 3 hours; it loses not more than 10 mg/g.
pH value (1.13). pH of a 100 mg/mL solution in carbon-dioxide-free water R, 5.5 to 6.5.
Related substances
Prepare fresh solutions and perform the tests without delay.
Carry out the test as described under 1.14.1 Chromatography, High-performance liquid chromatography using a stainless steel column (25 cm x 4.6 mm) packed with base deactivated particles of silica gel, the surface of which has been modified with chemically-bonded octadecylsilyl groups (5 µm).
The mobile phases for gradient elution consist of a mixture of mobile phase A and mobile phase B using the following conditions:
mobile phase A: 4 volumes of acetonitrile for chromatography R, 70 volumes of 0.02 mol/L sodium octanesulfonate R solution, 10 volumes of phosphate buffer pH 2.8 and 16 volumes of water R;
mobile phase B: 17 volumes of acetonitrile for chromatography R, 70 volumes of 0.02 mol/L sodium octanesulfonate R solution, 10 volumes of phosphate buffer pH 2.8 and 3 volumes of water R.
Prepare the sodium octanesulfonate solution by dissolving 4.7 g of sodium octanesulfonate R in 1000 mL of water R.
Prepare the phosphate buffer pH 2.8 by dissolving 27.2 g of potassium dihydrogen phosphate R in 800 mL of water R, adjust the pH to 2.8 by adding phosphoric acid (~20 g/L) TS and dilute to 1000 mL with water R.
|
Time (min) |
Mobile phase A (% v/v) |
Mobile phase B (% v/v) |
Comments |
|
0–16 |
100 |
0 |
Isocratic |
|
16–18 |
100 to 0 |
0 to 100 |
Linear gradient |
|
18–28 |
0 |
100 |
Isocratic |
|
28–30 |
0 to 100 |
100 to 0 |
Return to initial composition |
|
30–45 |
100 |
0 |
Re-equilibration |
Prepare the following solutions in mobile phase A. For solution (1) use a solution containing 1.0 mg of the test substance per mL. For solution (2) dilute a suitable volume of solution (1) to obtain a concentration equivalent to 5.0 μg of cycloserine per mL. For solution (3) use a solution containing 10.0 µg of D-serine R per mL. For solution (4) dilute a suitable volume of solution (3) to obtain a concentration of 2.0 µg of D-serine R per mL. For solution (5) dilute a suitable volume of solution (1) to obtain a concentration of 50 μg of cycloserine per mL. Heat this solution carefully in a boiling water-bath for 30 minutes. For solution (6) mix 1 mL of solution (3) with 1 mL of solution (5).
Operate with a flow rate of 1.0 mL per minute. As a detector use an ultraviolet spectrophotometer set at a wavelength of 195 nm.
Maintain the column temperature at 45 °C.
Inject 50 μL of solution (6). The test is not valid unless the peak corresponding to D-serine with a relative retention time of about 0.23 and the large degradation peak with a relative retention time of about 0.27 are baseline separated.
Inject alternatively 50 μL each of solutions (1), (2) and (4).
In the chromatogram obtained with solution (1) the following impurities, if present, are eluted at the following relative retention with reference to cycloserine (retention time about 14 minutes): impurity B (D-serine) about 0.23; impurity C about 0.35; and impurity A about 1.8. Use the chromatogram obtained with solution (4) to identify the peak due to impurity B.
In the chromatogram obtained with solution (1):
· the area of any peak corresponding to impurity B (D-serine) is not greater than the area of the principal peak in the chromatogram obtained with solution (4) (0.2%);
· the area of any other impurity peak is not greater than the area of the principal peak in the chromatogram obtained with solution (2) (0.5%);
· the sum of the areas of all impurity peaks, other than any peak corresponding to impurity B (D-serine), is not greater than three times the area of the principal peak in the chromatogram obtained with solution (2) (1.5%). Disregard any peak with an area less than 0.1 times the area of the principal peak in the chromatogram obtained with solution (2) (0.05%).
Assay
Dissolve about 0.1 g, accurately weighed, in 5 mL of water R. Add 75 mL of 2-propanol R and titrate with carbonate-free sodium hydroxide (0.1 mol/L) VS using thymolphthalein/ ethanol TS as indicator. Perform a blank determination and make any necessary correction.
Each mL of sodium hydroxide (0.1 mol/L) VS is equivalent to 10.21 mg of C3H6N2O2.
Impurities
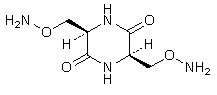
A. (3R,6R)-3,6-bis[(aminooxy)methyl]piperazine-2,5-dione (cycloserine dimer),
B. (2R)-2-amino-3-hydroxypropanoic acid (D-serine),
C. condensation product with unknown structure.