Monographs: Pharmaceutical substances: Dacarbazine (Dacarbazinum)
Molecular formula. C6H10N6O
Relative molecular mass. 182.2
Graphic formula
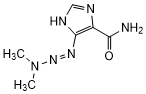
Chemical name. 5-(3,3-dimethyltriaz-1-en-1-yl)-1H-imidazole-4-carboxamide; CAS Reg. No. 4342-03-4.
Description. A colourless or pale yellow, crystalline powder.
Solubility. Slightly soluble in water R and ethanol (~750 g/L) TS, practically insoluble in dichloromethane R.
Category. Cytotoxic drug.
Storage. Dacarbazine should be kept in a tightly closed container, protected from light, and stored at a temperature not exceeding 8 °C.
Additional information. CAUTION: Dacarbazine must be handled with care, avoiding contact with the skin and inhalation of airborne particles.
Requirements
Dacarbazine contains not less than 98.5% and not more than 101.0% of C6H10N6O, calculated with reference to the anhydrous substance.
Identity tests
- Either test A alone or tests B and C may be applied.
A. Carry out the examination as described under 1.7 Spectrophotometry in the infrared region. The infrared absorption spectrum is concordant with the spectrum obtained from dacarbazine RS or with the reference spectrum of dacarbazine.
B. The absorption spectrum (1.6) of a 6 μg/mL solution in hydrochloric acid (~4 g/L) TS, when observed between 200 nm and 400 nm, exhibits a maximum at about 323 nm and a pronounced shoulder at 275 nm.
C. Carry out the test as described under 1.14.1 Chromatography, Thin-layer chromatography using silica gel R6 as the coating substance and a mixture of glacial acetic acid R, water R and butanol R (1:2:5 V/V/V) as the mobile phase. Apply separately to the plate 10 µL of each of the following 2 solutions in methanol R: containing (A) 0.4 mg of the test substance per mL and (B) 0.4 mg of dacarbazine RS per mL. Develop the plate for a distance of 15 cm. After removing the plate from the chromatographic chamber allow it to dry in air or in a current of air. Examine the chromatogram under ultraviolet light (254 nm).
The principal spot in the chromatogram obtained with solution (A) corresponds in position, appearance and intensity with the spot due to dacarbazine in the chromatogram obtained with solution (B).
Clarity and colour of solution. Dissolve 0.25 g of the test substance in a 210 g/L solution of citric acid R and dilute to 25.0 mL with the same solution. The solution is clear and not more intensely coloured than reference solution BY6, when analysed as described under 1.11.2 Degree of coloration of liquids, Method II.
Sulfated ash (2.3). Not more than 1.0 mg/g.
Water. Determine as described under 2.8 Determination of water by the Karl Fischer method, method A. Use 1.00 g of the test substance. The water content is not more than 5 mg/g.
Impurity D. Carry out the test as described under 1.14.1 Chromatography, Gas chromatography with an apparatus equipped with an injection system for the performance of static head-space chromatography. Use a fused-silica capillary column 30 m long and 0.53 mm in internal diameter coated with base-deactivated polyethyleneglycol R (film thickness: 1.0 µm).
As a detector use a flame ionization detector.
Use helium for chromatography R as the carrier gas with a flow rate of 13 mL/min.
Use a split ratio of 1:1.
The following head-space injection conditions may be used:
|
Equilibration temperature (°C) |
60 |
|
Equilibration time (minutes) |
10 |
|
Transfer line temperature (°C) |
90 |
|
Pressurization time (seconds) |
30 |
|
Injection volume (mL) |
1 |
Maintain the temperature of the column at 35°C for 3 minutes, then raise the temperature within 8 minutes to 165 °C, maintaining the temperature of the injection port at 180 °C and that of the flame ionization detector at 220 °C.
Prepare the following solutions. For solution (A) dilute 1.00 g of dimethylamine R (impurity D) to 100.0 mL with water R. For solution (C) dilute 1.00 g of trimethylamine R to 100.0 mL with water R.
Prepare the following vials. For vial (1) transfer 0.200 g of the test substance into a 20 mL headspace vial and firmly attach the septum and cap. Using a 10 µL syringe, inject 5 µL of water R into the vial. For vial (2) firmly attach the septum and cap to a 20 mL vial. Using a 10 µL syringe, inject 10 µL of solution (A) into the vial. For vial (3) firmly attach the septum and cap to a 20 mL vial. Using a 10 µL syringe, inject 10 µL of solution (A) and 10 µL of solution (B) into the vial.
Analyse vial (3). The test is not valid unless the resolution between the peaks due to impurity D and trimethylamine is at least 2.5.
Analyse alternately vial (1) and (2).
In the chromatogram obtained with vial (1):
- the area of any peak corresponding to impurity D is not greater than the area of the corresponding peak in the chromatogram obtained with vial (2) (0.05%).
Related substances
Use freshly prepared solutions and protect them from light.
- Perform test A and B.
A. Carry out the test as described under 1.14.1 Chromatography, High-performance liquid chromatography using a stainless steel column (25 cm x 4.6 mm) packed with particles of silica gel, the surface of which has been modified with chemically-bonded octadecylsilyl groups (5 µm).
Prepare the mobile phase by dissolving 15.63 g of glacial acetic acid R in a solution containing 2.33 g of docusate sodium R per L of water R and dilute to 1000 mL with the same solvent. Prepare the mobile phase freshly every day and flush the column with a mixture of equal volumes of methanol R and water R after all tests have been completed or at the end of the day, for at least 2 hours.
Operate with a flow rate of 1.2 mL per minute. As a detector use an ultraviolet spectrophotometer set at a wavelength of 254 nm.
Prepare the following solutions in distilled water R. For solution (1) dissolve 50.0 mg of the test substance and 75 mg of citric acid R and dilute to 5.0 mL. For solution (2) dissolve 5.0 mg of dacarbazine impurity A RS and dilute to 50.0 mL. Dilute 5.0 mL of this solution to 25.0 mL.
Inject alternately 25 μL of each of solution (1) and (2). Record the chromatograms for 3 times the retention time of impurity A (retention time about 3 minutes).
In the chromatogram obtained with solution (1):
- the area of any peak corresponding to impurity A is not greater than the area of the corresponding peak in the chromatogram obtained with solution (2) (0.2%);
- the area of any other impurity peak eluting after impurity A is not greater than 0.5 times the area of the peak due to impurity A in the chromatogram obtained with solution (2) (0.10%).
B. Carry out the test as described under 1.14.1 Chromatography, High-performance liquid chromatography using the conditions given above under test A with the following modifications.
Prepare the mobile phase by mixing 45 volumes of the mobile phase described under test A with 55 volumes of methanol R.
Prepare the following additional solution in distilled water R. For solution (3) dissolve 5.0 mg of dacarbazine impurity B RS, add 0.5 mL of solution (1) and dilute to 10.0 mL. Dilute 1.0 mL of this solution to 50.0 mL.
Inject alternately 10 μL of each of solution (1) and (3). Record the chromatograms for twice the retention time of dacarbazine (retention time about 12 minutes). The test is not valid unless the resolution between the peaks due to impurity B (with a relative retention of about 0.7) and dacarbazine is at least 1.5.
In the chromatogram obtained with solution (1):
- the area of any peak corresponding to impurity B is not greater than the area of the corresponding peak in the chromatogram obtained with solution (3) (0.1%);
- the area of any other impurity peak is not greater than the area of the peak due to dacarbazine in the chromatogram obtained with solution (3) (0.10%);
- the sum of the areas of all impurities peaks is not greater than 5 times the area of the peak due to darcarbazine in the chromatogram obtained with solution (3) (0.5%). Disregard any peak with an area less than 0.5 times the area of the peak due to dacarbazine in the chromatogram obtained with solution (3) (0.05%).
Assay
Protect the solutions from light throughout the assay.
Dissolve 0.150 g in 30 mL of anhydrous acetic acid R. Titrate with perchloric acid (0.1 mol/L) VS, determining the end-point potentiometrically. Each mL of perchloric acid (0.1 mol/L) VS is equivalent to 18.22 mg of C6H10N6O.
Impurities
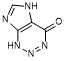
A. 1,5-dihydro-4H-imidazo[4,5-d]-1,2,3-triazin-4-one (2-azahypoxanthine) (degradation product)
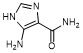
B. 5-amino-1H-imidazole-4-carboxamide (synthesis-related impurity)
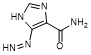
C. 5-diazenyl-1H-imidazole-4-carboxamide
![]()
D. N-methylmethanamine (dimethylamine)